For our international customers, please be advised that orders cannot be placed through our website by customers in countries with International Distributor representation.
Galactosidase, Beta - Manual
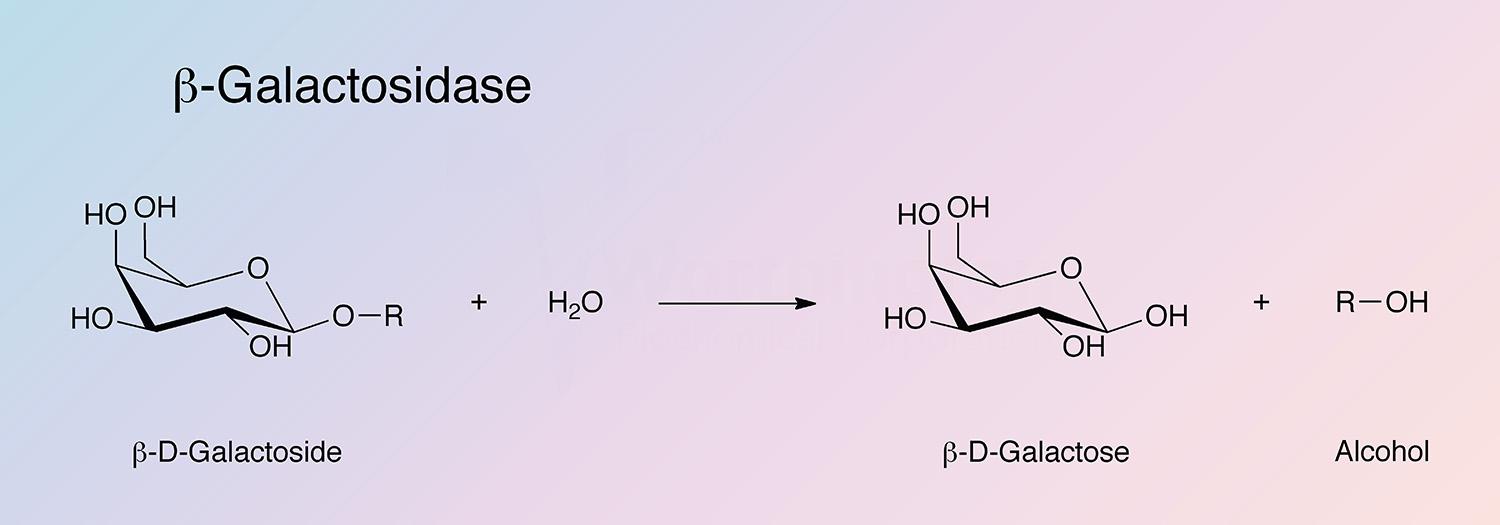
Beta-galactosidase (Lactase) catalyzes the following hydrolysis:
β-Galactosidases are widespread, in microorganisms, animals and plants. That from the Escherichia coli strain K12 has been particularly studied at Anfinsen's laboratory in connection with genetic experiments on gene regulation of protein synthesis. Craven, Steers and Anfinsen 1965). The enzyme has been reviewed in detail by Wallenfels and Weil (1972). Lactase may be used as a reagent for determining lactose in blood and other biological fluids.
Another important application is in food processing. Of special interest is its use in the treatment of milk to meet the needs of the large percentage of the world population afflicted with lactose intolerance. Industrial applications necessitate the enzyme's immobilization on which considerable investigation has been reported (Byrne and Johnson 1975; Narinesingh et al. 1975; Paine and Carbonell 1975; Faulsitch et al. 1974; Bunting and Laidler 1972 , Lilly 1971, Khare and Gupta 1988, and Park and Hoffman 1990).
Characteristics of β-Galactosidase from E. coli:
A review of substrate requirements is offered by Wallenfels and Malhotra (1960).
The enzyme is tetrameric, being composed of four identical subunits of 135,000 daltons, each with an active site which may be independently active (Melcher and Messe 1973). The enzyme is readily fragmented into small peptides (Marinkovic et al. 1975). The amino acid analysis indicates approximately 1170 residues per subunit (Fowler and Zabin 1970). See also Langley et al. (1975) and Naider et al. (1972). Kaneshiro et al. (1975) report on an active dimer.
Contaxis and Reithel (1974) report a small molecule with high 260 nm absorbance which is bound to the enzyme in crude extracts as a stabilizer. On adding it to the pure enzyme, activity and heat stability are enhanced.
Bronskil and Wong (1971) suggest that the ribosomal enzyme may be an intermediate stage in the assembly of the quarternary structure.
540,000 (Steers et al. 1965; Cohen and Mire 1971; Contaxis and Reithel 1971).
6 - 8. See also Tenu et al. (1971 and 1972).
![]() = 20.9.
= 20.9.
In the absence of Mg2+ ions, β-mercaptoethanol causes dissociation (Shifrin et al. 1970. See also Case et al. (1973) and Loontiens et al. (1970).
The enzyme is stable 4 - 6 months when stored at 5°C.
The reaction probably involves a galactosyl-enzyme intermediate (van de Groen 1973). See also Hill and Huber (1971) and Sinnott and Souchard (1973). Hartl and Hall (1974) report a second β-galactosidase without lactase activity in vivo but with high ONPG (o-nitrophenyl, β-D-galactopyranoside) activity. Monovalent cations have a stimulatory effect on the enzyme (Becker and Evans 1969). The alcohols, methanol, ethanol, i-propanol, and n-propanol, at 5% concentration, all increase the rate of o-nitrophenyl, β-D-galactopyranoside cleavage (Shifrin and Hunn 1969). The enzyme is protected against heat-inactivation by 5-phosphorylribose 1-pyrophosphate in the presence of β-mercaptoethanol (Moses and Sharp 1970).