For our international customers, please be advised that orders cannot be placed through our website by customers in countries with International Distributor representation.
Creatine Kinase - Manual
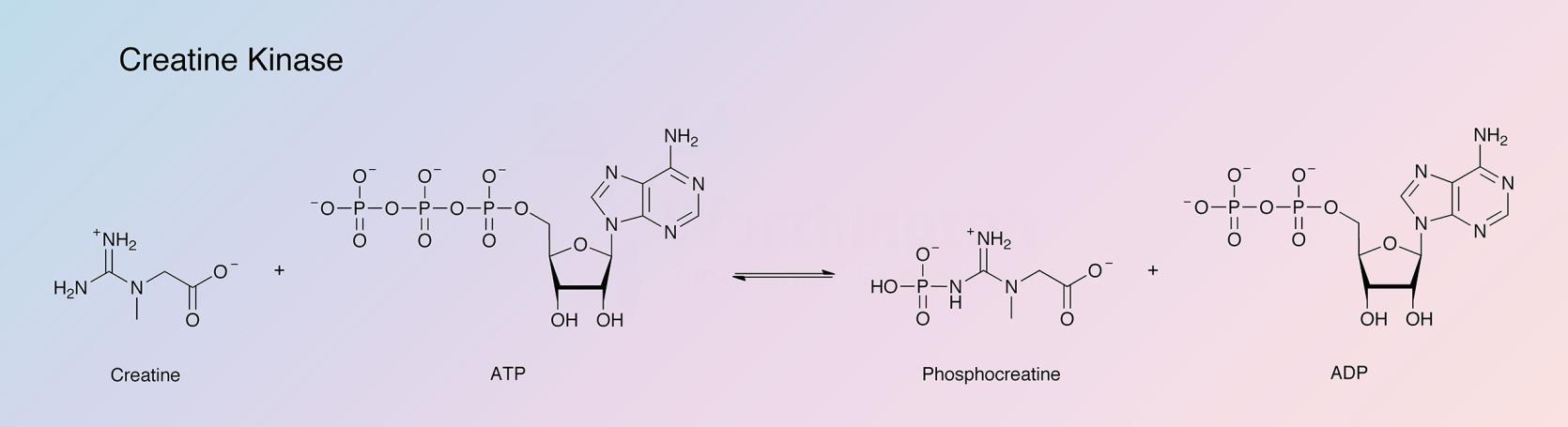
Creatine kinase catalyzes the following reaction:
Creatine kinase (CK) is widely distributed and seems to be primarily concerned with ATP regeneration. According to Watts (1973) CK may represent 10-20% (w/v) of muscle cytoplasmic protein.
The enzyme is dimeric and exists as three isozymes: MM (muscle), MB (hybrid), and BB (brain). Names indicate major tissue of origin. Since the MB isozyme has its highest concentration in heart muscle, its level in the serum has diagnostic value.
Most investigational work has been on the soluble enzyme from rabbit muscle. (There is also an insoluble CK associated with mitochondria).
Characteristics of Creatine Kinase from Rabbit Muscle:
Besides creatine (N-methylglycocyamine) only N-ethylglycocyamine and glycocyamine act as phosphoryl acceptors. Creatinine, D- and L-arginine, histidine, and taurocyamine are inactive (Kuby and Noltmann 1962).
The enzyme is dimeric, its identical subunits will combine with others from different species to form active hybrids (Dawson et al. 1967). The amino acid composition has been determined (Watts 1973). Active center studies have been reported by Borders and Riordan (1975); James and Cohn (1974) and Buechter et al. (1992). Smith and Kenyon (1974) question whether the "active" SH group of CK is, in fact, essential. See also McLaughlin (1974) and McLaughlin et al. (1972) and Roustan et al. (1973). O'Sullivan et al. (1972) report on ADP analog binding.
81,000 (Kuby and Noltmann 1962).
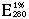 = 8.76 (Kuby et al. 1954).
= 8.76 (Kuby et al. 1954).
Various sulfhydryl reagents, chelating agents, some adenosine phosphate compounds, orthophosphate, pyro- and tripolyphosphate, adenosine, Cl-, SO42-, acetate (slight), and other compounds such as dibenamine, phenothiazone, and 3,5-dinitro-o-cresol. ADP strongly inhibits the forward reaction competitively with respect to ATP and noncompetitively with respect to creatine (Kuby and Noltmann 1962). Creatinine phosphate acts as competitive inhibitor with respect to phosphocreatine (Gercken and Døring 1974).
If kept dry, the refrigerated lyophilized preparation is stable for 6-12 months.
Engelborghs et al. (1975) have reported on initial product formation, and Gercken and Döring (1974) show that creatinine phosphate is not a substrate but is a competitive inhibitor to creatine phosphate. Various divalent cations such Mg2+, Ca2+, and Mn2+ activate the enzyme.
Reducing agents such as cysteine and thioglycolate.