For our international customers, please be advised that orders cannot be placed through our website by customers in countries with International Distributor representation.
DNA Polymerase, Taq - Manual
Taq DNA polymerase is a DNA-dependent polymerase. Taq refers to Thermus aquaticus, the bacteria from which this thermostable polymerase was first isolated. The enzyme exhibits optimal activity at 75°C.
DNA polymerase I was the first enzyme thought to be involved in DNA synthesis and was the only DNA polymerase to be studied from the late 1950s to the early 1970s (Kornberg et al. 1956, Bessman et al. 1957, Buttin and Kornberg 1966, Englund et al. 1968, Deutsch and Kornberg 1969, Englund et al. 1969, and Kornberg et al. 1974). In 1969, DeLucia and Cairns isolated an E. coli strain with a mutation that affected the DNA polymerase and surprisingly found that the mutant synthesized DNA normally. This discovery casted doubts on the role of DNA polymerase in replication and led groups to search for other replication enzymes.
DNA polymerase II and III of E. coli were soon isolated and characterized (Kornberg and Gefter 1972, and Livingston and Richardson 1975). However, few attempts were made to isolate DNA polymerase from thermophiles (Stenesh and Roe 1972). In 1976, DNA polymerase from the thermophilic bacterium Thermus aquaticus was first isolated and purified, and its remarkable stability at high temperatures was described (Chien et al. 1976).
Soon after its purification, Taq DNA polymerase was found to be highly useful in the polymerase chain reaction (PCR) method of DNA amplification. Compared to E. coli DNA polymerase I, Taq polymerase’s high specificity of primer binding at elevated temperatures was found to give a higher yield of the desired product with less non-specific amplification product (Saiki et al. 1985, Mullis and Faloona 1987, and Lawyer et al. 1989). This feature paved the way for dramatic improvements to the PCR method. Since its optimal temperature is so high, Taq polymerase only needs to be added to a PCR reaction at the beginning, rather than before each round of amplification.
The crystal structure was determined by Kim et al. in 1995. Recent work with Taq polymerase has included developing inhibitor-resistant mutants of Taq polymerases for use in PCR assays (Baar et al. 2011) and investigating the molecular basis of substrate specificity through thermodynamic and structural studies (Obeid et al. 2010, and Wowor et al. 2010).
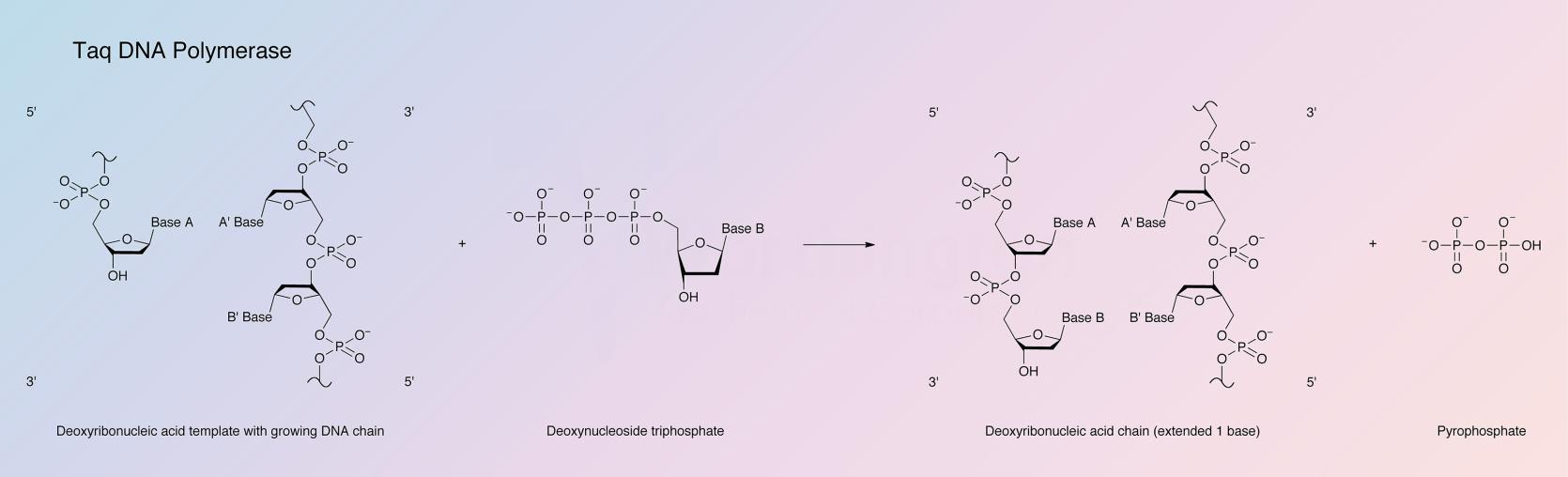
DNA synthesis proceeds in the 5’ to 3’ direction and requires the presence of dNTPs and primer template DNA. The similar structure of DNA polymerases has indicated that these enzymes use an identical two metal ion-catalyzed polymerase mechanism (Beese and Steitz 1991). One metal ion activates the primer’s 3’-OH for attack on the a-phosphate of the dNTP. The other metal ion stabilizes the negative charge of the leaving oxygen and chelates the b- and g-phosphates (Steitz 1999).
The enzyme is thermostable and exhibits optimal activity at 75°C. It is stable at -20°C when undiluted. If needed, it is recommended to dilute the enzyme in storage buffer containing 0.1% nuclease-free BSA (Worthington Code:BSANF).
The enzyme consists of a single polypeptide chain. The domain shape of all polymerases whose structures are known has been described as a “right hand” with “thumb”, “palm”, and “finger” domains (Kohlstaedt et al. 1992). The palm region is thought to catalyze the phosphoryl transfer, and the finger region is thought to interact with the incoming nucleoside triphosphate and the template base it is paired to. The thumb is believed to help in positioning the DNA and in translocation (Brautigam and Steitz 1998).
Taq polymerase displays little, if any, 3’to 5’ exonuclease activity. It is possible that mutations, deletions, or insertions destroyed this activity; E. coli polymerase I displays little sequence similarity in the region believed to be the 3’ to 5’ exonuclease region. Six amino acid residues believed to be essential for polymerase activity in DNA polymerase I include Met-512, Arg-682, Lys-758, Tyr-766, Arg-841, and His-881 (Lawyer et al. 1989). All residues except Met-512 are conserved in Taq polymerase.
Thermophilic organisms have been found to have high GC content. E. coli polymerase has been shown to contain 52.0% GC conent, whereas the gene for Taq polymerase contains 67.9% (Lawyer et al. 1989).
- Polymerase chain reaction (PCR)
- Primer dependent DNA labeling or DNA sequencing (particularly since the reaction can proceed at high temperature)
P19821
94 kDa (gel filtration, Lawyer et al. 1993)
- Active from 7.5-9.5, around 9 is optimal (20°C)
- 6.03 (Theoretical)
- 110,380 cm-1 M-1 (Theoretical)
- E1%, 280 = 11.75 (Theoretical)
- Bromophenol blue (Wittwer and Garling 1991)
- Aptamer (Yakimovich et al. 2003)
- Pyranicin (Takahashi et al. 2008)
- Catalpol (Pungitore et al. 2004)
- KCl (greater than [75 mM])
- Urea, DMSO, DMF, formamide, and SDS (Wittwer and Garling 1991)