For our international customers, please be advised that orders cannot be placed through our website by customers in countries with International Distributor representation.
Glucosidase, Beta - Manual
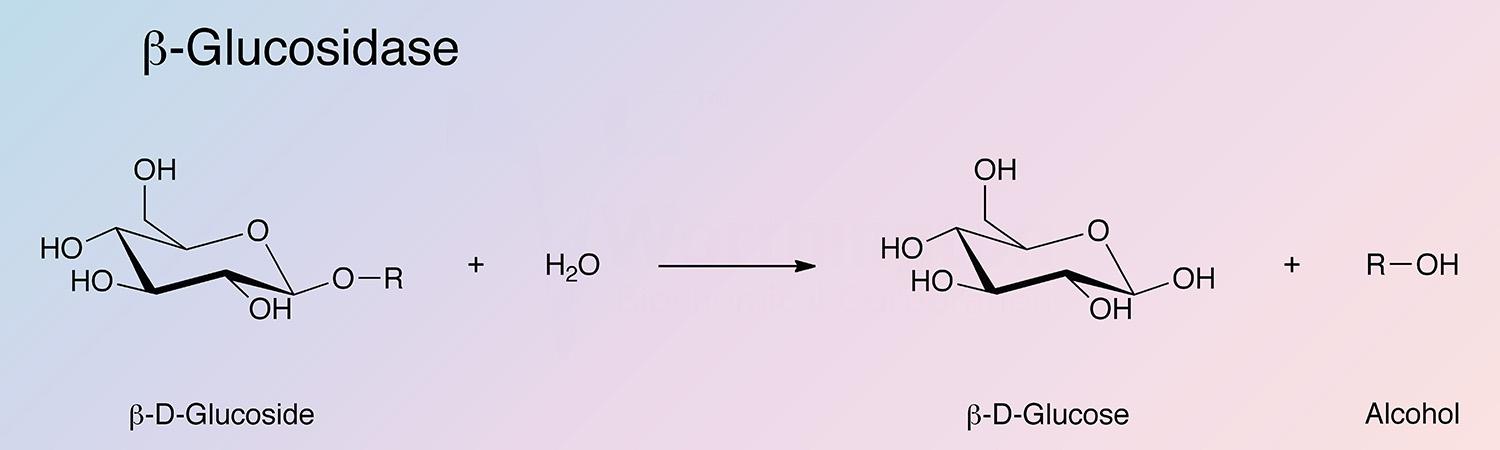
β-Glucosidases catalyze the hydrolysis of glucosides:
Emulsin originally was the term applied to the extract of sweet almonds. It remains as the historical prototype of β-glucosidase. Many other sources of the enzyme have been described. Emulsin also shows significant D-galactosidase activity and there is some question as to whether or not it is one enzyme with two activities, because non-parallel response to inhibitors points to two separate enzymes. D-mannosidase has also been reported in almond emulsin (Schwartz et al. 1970). The enzyme has been reviewed by Veibel (1950), Larner (1960), and Nisizawa and Hashimoto (1970).
Characteristics of β-Glucosidase from Almond:
β-glucosides and β-galactosides are reportedly substrates for the enzyme (Kelemen and Whelan 1966). β-D-fucosides are also attacked (Conchie et al. 1967). This is questioned by Schwartz et al. (1970) who offer data supporting the existence of separate enzymes.
2 active components, 117,000 and 66,500 (Helferich and Kleinschmidt 1965).
Butyryl glycoside in acetate, pH 4.4 (Veible 1950), p-nitrophenyl glycoside 5.2 - 6.0 (Schwartz et al. 1970).
HgCl2, other heavy metal ions, sulfhydryl-binding compounds and polyols.
The enzyme is stable for 1 - 2 years at 2-8°C.
There has been interest in the mammalian form of the enzyme, particularly in relation to Gaucher's disease. Losman (1974) reported that an iso-enzyme of β-glucosidase is lacking in the lymphocytes of homozygotes and a significant decrease is found in heterozygotes. Spleen and liver enzyme activity was lowered in Gaucher's patients when assayed with the synthetic substrate, 4-methylumbelliferal β-D-glycoside.
Km (M) Glu-I 0.072 (Schwartz et al 1970.)
Glu-II 0.080 (Schwartz et al. 1970)