For our international customers, please be advised that orders cannot be placed through our website by customers in countries with International Distributor representation.
Glucose Oxidase - Manual
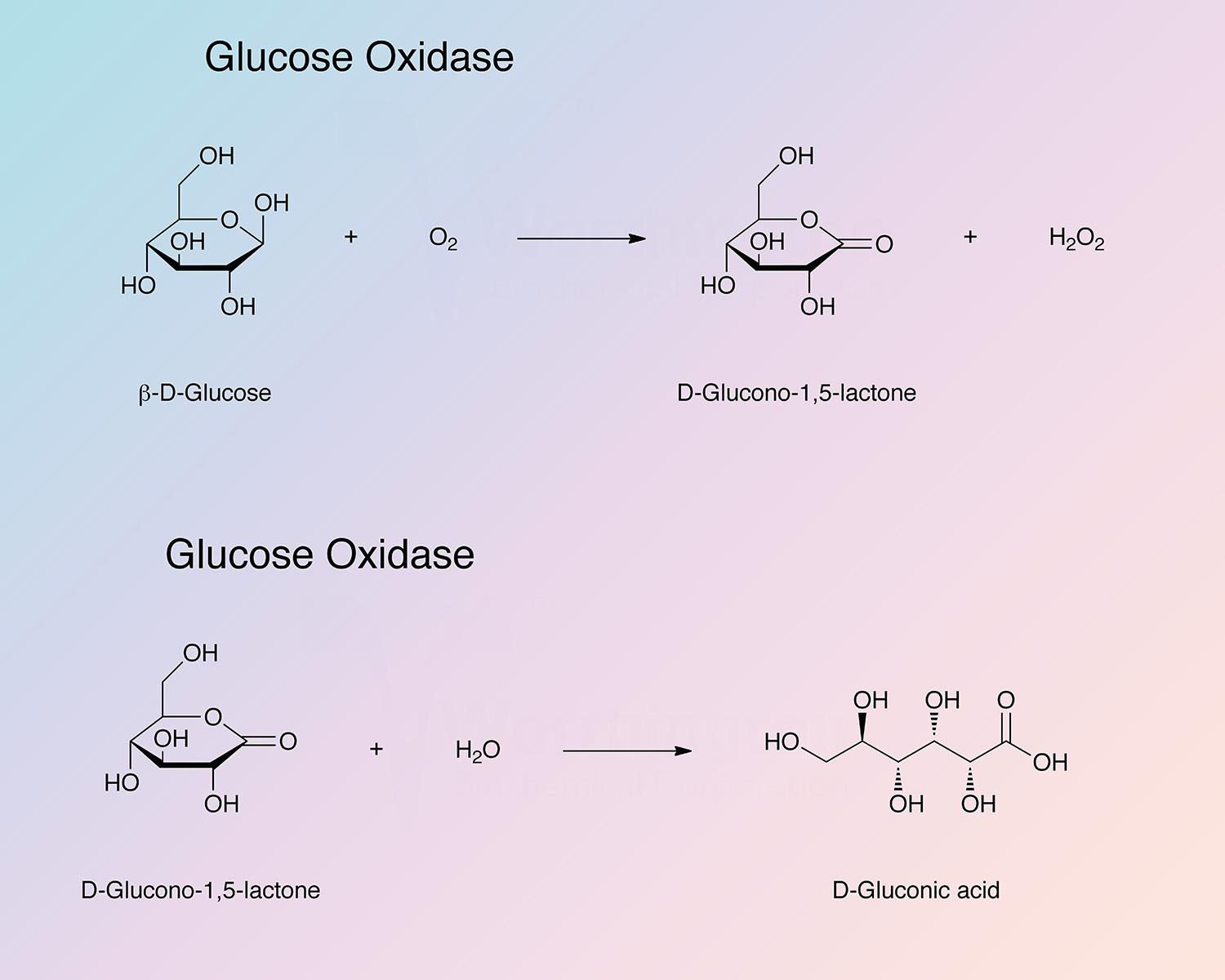
Glucose oxidase, a flavoenzyme, catalyzes the following reaction:
Bright and Porter (1975) have reviewed the kinetic behavior and redox states of the flavin coenzyme. Bentley (1963) has reviewed the general properties of the enzyme.
Since its discovery as an "antibiotic" (shown subsequently to be due to peroxide formation) there has been an interest in glucose oxidase, chiefly because of its utility in glucose estimation. Following Keston's report in 1956 of coupling the reaction to peroxidase and a chromogen, (qualitative) glucose "dip-sticks" became available for screening for urine glucose. Based on Teller's paper in the same year, Worthington offered the first quantitative enzymatic system for the colorimetric determination of glucose. For most clinical work the crude form of the A. niger enzyme has been satisfactory. However, it contains trace amount of polysaccharidases such as amylase, maltase, and sucrase which can contribute to falsely high glucose levels. The purified enzyme is free of these traces and is recommended for analytical use in the presence of di- or polysaccharides. Reports on glucose analytical systems employing the enzyme include: Williams et al. (1976), and Auses et al. (1975) wherein H2O2 produced reacts with Fe(CN) -- in the presence of luminol to produce luminescence proportional to the initial glucose concentration. See also Lott and Turner (1975).
Considerable work has been done on immobilized glucose oxidase. Reports include the following: Greenfield et al. (1975), Greenfeld and Laurence (1975), Lahoda et al. (1975), Mell and Maloy (1975), Tran-Minh and Broun (1975), Kunz and Stastny (1974), Miwa and Okuda (1974), Weibel et al. (1973), Gestrelius et al. (1972), et al. (1972), Jemmali and Rodriquez-Kabana (1972), Okuda and Miwa (1971), Weibel and Bright (1971a), Hornby et al. (1970), Weetall and Hersh (1970).
Characteristics of Glucose Oxidase from A. niger:
The enzyme is highly specific for β-D-glucose. The α anomer is not acted upon. 2-deoxy-D-glucose, D-mannose and D-galactose exhibit low activities as substrate. (See Bentley 1966).
The enzyme consists of two identical polypeptide chain subunits (80,000 daltons) covalently linked by disulfide bonds (O'Malley and Weaver 1972). Each subunit contains one mole of Fe and one mole of FAD (flavin-adenine dinucleotide). Tsuge et al. (1975) report the molecule to be approximately 74% protein, 16% neutral sugar and 2% amino sugars. They indicate that the FAD is replaceable with FHD (flavin-hypoxanthine dinucleotide) without loss of activity.
160,000 (Tsuge et al. 1975).
5.5 with broad range 4 - 7 (Bright and Appleby 1969). See also Weibel and Bright (1971b).
Ag+, Hg2+, Cu2+ ( Nakamura and Ogura 1968). FAD binding is inhibited by several nucleotides (Swobada 1969). See also Rogers and Brandt (1971).
Dry preparations are stable for years when stored cold. Solutions are reasonably stable under a variety of conditions.