For our international customers, please be advised that orders cannot be placed through our website by customers in countries with International Distributor representation.
Lactoperoxidase - Manual
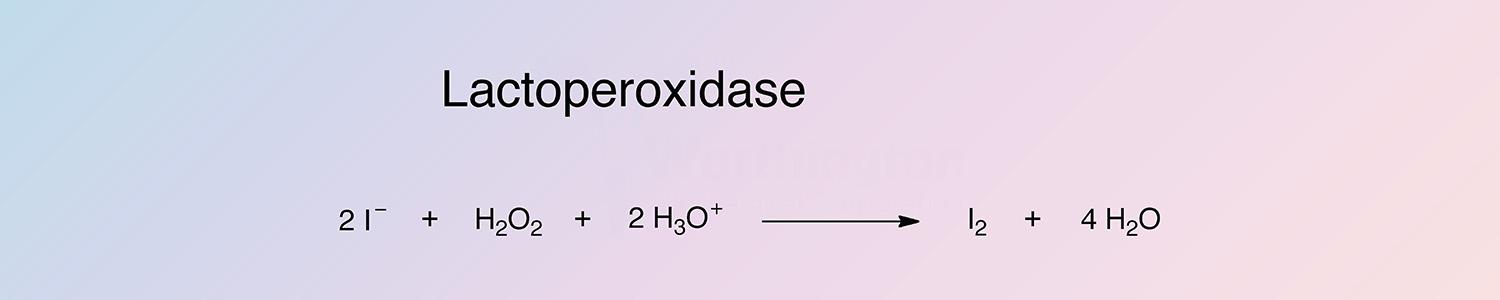
Lactoperoxidase (LPO) catalyzes the hydrogen peroxide oxidation of iodide.
The bovine milk enzyme is identical to that formed in bovine lacrimal and salivary glands (Morrison et al. 1965; and Morrison and Allen, 1963). It is possible that LPO may be important in controlling bacterial flora (Bjorck et al. 1970; Gothefors and Marklund, 1975; and Morrison and Allen, 1966). LPO is useful for labelling proteins with radioiodine (Gow and Wardlaw 1975; Holohan et al. 1973; Morrison and Bayse 1973; Bayse et al. 1972; Frantz and Turkington 1972; and Marchalonis 1969). For membrane studies the large LPO molecule limits labelling to the exposed surface. Poduslo and Braun (1975) report on the topography of membrane proteins of the myelin sheath. Shin and Carraway (1974) and Phillips and Morrison (1971) report on erythrocyte and Nachman et al. (1973) on platelet membranes. Haustein (1975) studied the cell-surface protein of lymphoma cells and Huber et al. (1975) mitochondrial membranes. Chloroplast membranes were reported on by Arntzen et al. (1974). Jone and Hager (1976), and Hogg (1974) compare membrane protein of normal and transformed cell. Immobilized LPO has also proven of interest (Johnson et al. 1975; Karonen et al. 1975; and David 1972).
Characteristics of Lactoperoxidase from Bovine Milk:
LPO is a glycoprotein with a single hemin prosthetic group per molecule (Hultquist and Morrison, 1963). It may consist of two isozymes (Rombauts et al. 1967; Morrison et al. 1957; and Polis and Schmukler, 1953). See also Morrison and Hultquist (1963).
77,500 (Rombauts et al. 1967).
![]() = 13.9.
= 13.9.
LPO is inhibited by hydrazines (Allison et al. 1973). Dolman et al. (1968) report on the kinetics of cyanide inhibition.
LPO like other peroxidases, in the presence of H2O2 catalyzes the oxidation of many phenols and aromatic amines (pyrogallol, ascorbate, guiacol, etc.). See Morrison (1970) page 657 for a comparison of specific activity of a variety of peroxidases, also Maguire and Dunford (1971). Morrison and Bayse (1973) indicate that the iodide reacts directly with the heme group, the complex then iodinates the substrate on adding H2O2. Bayse et al. (1972) and Morrison and Bayse (1970) report on iodination kinetics. Chung and Wood (1970) report on the oxidation of thiocyanate and Maguire and Dunford (1972) on that of iodide.
A412/A280 = 0.93-0.96 Allen and Morrison 1963).