For our international customers, please be advised that orders cannot be placed through our website by customers in countries with International Distributor representation.
Luciferase - Manual
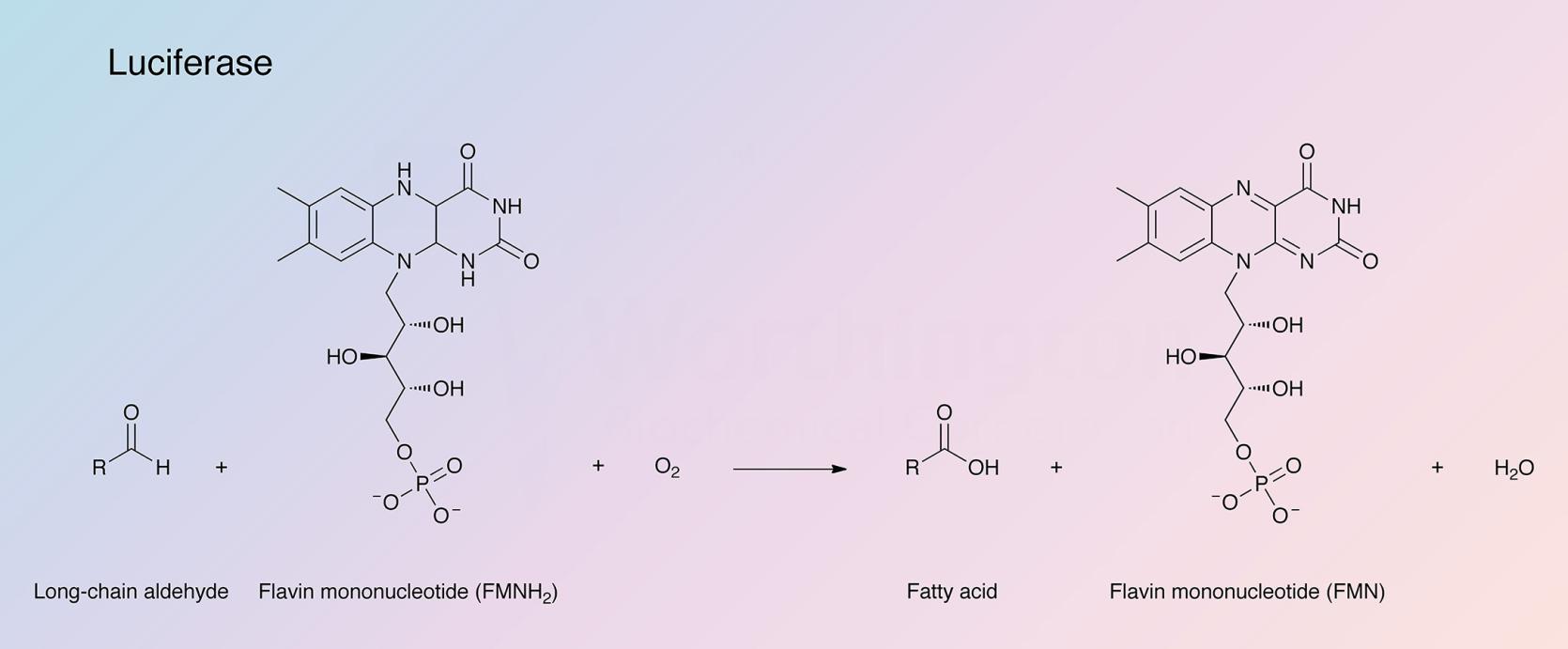
Bacterial luciferase (LU) catalyzes the oxidation of reduced flavin mononucleotide (FMNH2) and a long chain aldehyde by molecular oxygen to yield FMN, the corresponding acid, H2O and light. (Baldwin et al. 1975; Balny and Hastings 1975; Becvar and Hastings 1975).
See also Hastings et al. (1966); the aldehyde is not essential to the reaction but has a striking effect upon the luminescence. The FMN can be reduced by molecular hydrogen with Pt or Pd catalyst, or by treatment with dithionite, but because FMNH2 is so rapidly oxidized by air, the use of NADH is a more workable laboratory method and more useful in analytical applications. The mechanism by which bacterial luciferase functions to produce light was studied by Dr. J. Woodland Hastings' group. In an article describing partial and rapid purification of LU using affinity chromatography, Waters et al. (1974) describe the specific activity of highly purified luciferase as being 1.4 X 1014 quanta per second per milligram. Brolin et al. (1971) developed a very sensitive assay for trace metabolic intermediates which can be involved in pyridine nucleotide dependent dehydrogenase reactions using bacterial luciferase. See also Chappelle and Picciolo (1971) for its use in assaying FMN and FAD. See Aflalo and DeLuca (1987) for its use monitoring ATP. A review of the use of firefly luciferase as a tool in molecular and cell biology is offered by Gould and Subramani (1988). A review of clinical applications for the enzyme is given by Kricka (1988).
Characteristics of Luciferase from Photobacterium fischerii:
Note: In Hastings' laboratory a strain of bacteria of uncertain origin, but with a different luciferase, was isolated and labelled MAV. A number of papers refer to the MAV enzyme. Hastings et al. (1969) have compared the two luciferases. The MAV strain has recently been identified as bacterium Beneckea harveyi (Tu et al. 1975).
The enzyme is specific for FMNH2. The aldehyde is not essential for the reaction (Hastings et al. 1966).
The enzyme is composed of two distinct subunits, a and b, each of approximately 40,000 molecular weight. Their amino acid content has been reported (Hastings et al. 1969). See also Meighen et al. (1970, 1971).
The luciferase α-β dimer has but one reduced flavin binding site (Becvar and Hastings 1975). Nicoli et al. (1974) and Nicoli and Hastings (1974) report a sulfhydryl group near the active center of MAV luciferase. See also Cline and Hastings (1972) and Gunsalus-Miguel et al. (1972).
Approximately 80,000 (Hastings et al. 1969).
6.8 (Hastings et al. 1969).
Oxidase inhibitors such as SKF (2-diethyl-aminoethyl-2,2-diphenyl valerate) are effective at concentrations as low as 10-5-10-4 M. (Nealson and Hastings 1972). p-Chloromercuribenzoic acid and riboflavin are both inhibitors.