For our international customers, please be advised that orders cannot be placed through our website by customers in countries with International Distributor representation.
Maltase - Manual
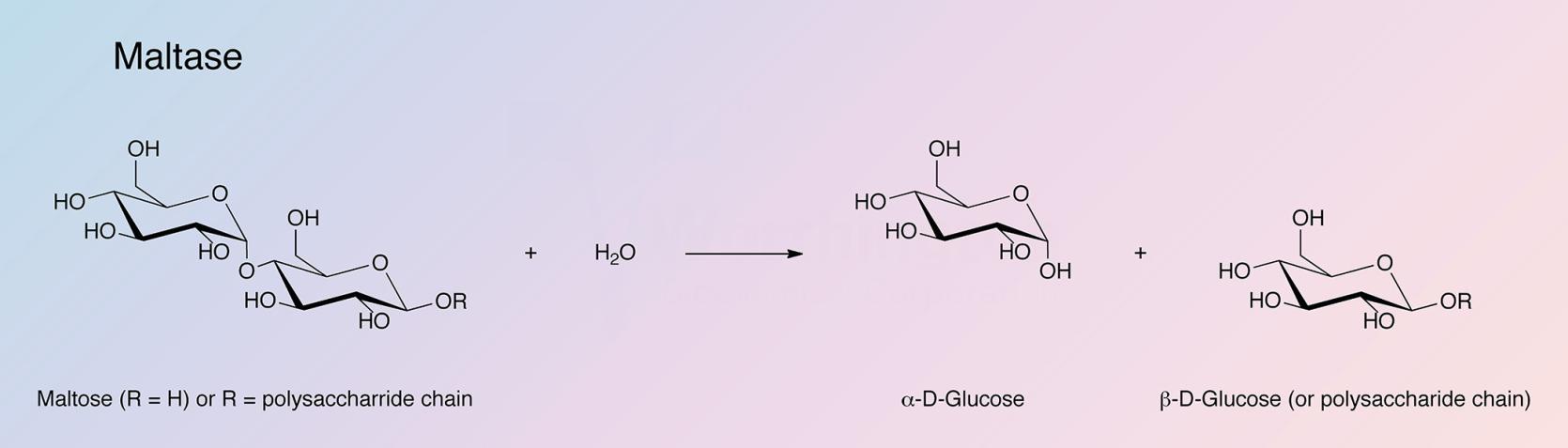
α-Glucosidase (Maltase) hydrolyzes the terminal, non-reducing 1,4-linked α-D-glucose residues with release of α-D-glucose. α 1,2 and α 1,3 bonds are cleaved at a much slower rate. The rate of hydrolysis decreases substantially with increasing substrate size. α-glucosidase will cleave sucrose but will not hydrolyze α-D-glucosides such as cellobiose.
Maltase, located in the brush border membrane of the enterocytes, was first separated in homogenates of pig intestinal mucosa and later in human preparations and exhibits quite complicated cross-specificity for different substrates. (Dahlqvist 1984).
Characteristics of Maltase from Baker's Yeast:
68,500 daltons.
7.0 - 7.5 using maltose as the substrate.
Thiol blocking compounds, heavy metal ions, histidine, and certain amines. Tris should not be used as a buffer due to its inhibitory effect.
Store at 2-8° C.