For our international customers, please be advised that orders cannot be placed through our website by customers in countries with International Distributor representation.
Malate Dehydrogenase - Manual
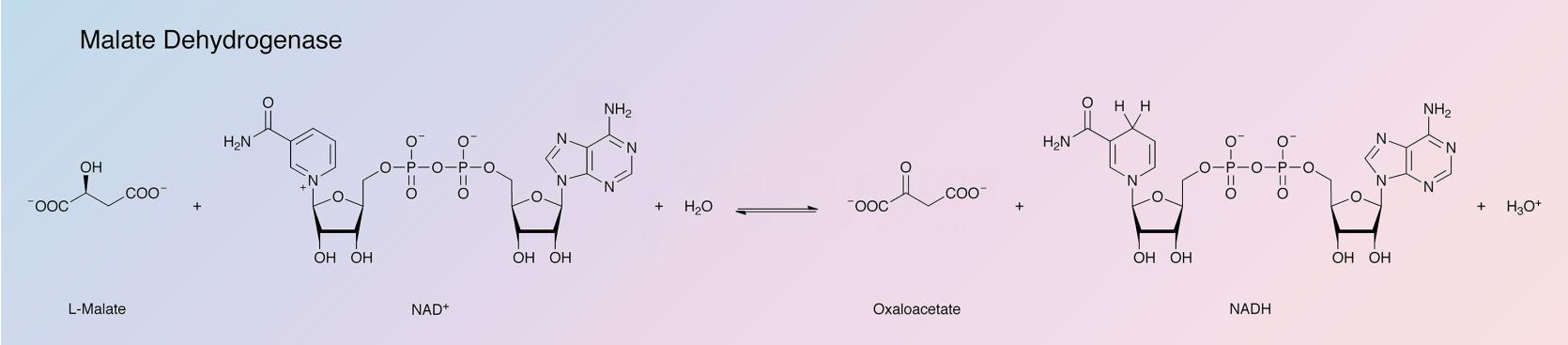
Malate dehydrogenase (MDH) catalyzes the interconversion of L-malate and oxaloacetate using nicotinamide adenine dinucleotide (NAD) as a coenzyme.
MDH is found in all eukaryotic cells as two isozymes: mitochondrial (m-MDH) and cytoplasmic (soluble, s-MDH). Prokaryotes contain only a single form. Pig heart MDH has been extensively studied. The two isozymes, both consisting of two very similar subunits of about 35,000 daltons and having similar enzymatic activity appear as different proteins (Bleile et al. 1975). Noyes et al. (1974) report on the structural similarity of mitochondrial MDH to L-3-hydroxyacyl CoA dehydrogenase (the cytoplasmic MDH being somewhat similar to lactate dehydrogenase e). Schindler (1975) indicates differences in binding sites. Banaszak and Bradshaw (1975) have reviewed the enzymes and have compared the amino acid compositions of the two isozymes from pig heart with those of other species. They suggest that the subforms of the isozymes that have been reported may be preparative artifacts. A number of non-mammalian malate dehydrogenases have been included in the references.
MDH is of interest to the clinician in that its activity in serum and cerebral/spinal fluid has been shown to be of diagnostic significance (Sharpe et al. 1973). See also Schwartz (1973).
Characteristics of Malate Dehydrogenase from Pig Heart (mitochondrial):
The heart enzyme is specific for L-malic acid, but Hayashi et al. (1966) report a D-malate dehydrogenase in Serratia marcescens. Thorne (1962) reports that m-MDH from ox heart, rat liver, rabbit kidney, and those from acetone powders of horse, pig and pigeon heart are similar in specificity and Michaelis constants.
The molecule consists of two polypeptide chains. There are two coenzyme binding sites per 70,000 daltons (Eberhardt and Wolfe 1975). Active center studies include those of Chen and Engel (1975), Foster and Harrison (1974 and 1975), et al. (1975), Wimmer et al. (1975), Holbrook and Wolfe (1972). See also Codd (1972) who reports a possible binding site for FMN.
70,000 (Thorne and Kaplan 1963). According to Devenyi et al. (1966) the molecule is composed of similar subunits of molecular weight 35,000.
7.4 (NADH ---> NAD).
![]() = 2.8 (Thorne 1962).
= 2.8 (Thorne 1962).
Phosphate, arsenate, and zinc ions are stimulatory (Blonde et al. 1967). Mercuribenzoate has also been shown to be an activator at low concentrations (Silverstein and Sulebele 1970; Kuramitsu 1968).
Several iodinated agents, thyroxine, iodine, cyanide, and molecular iodine also inactivate the enzyme by oxidizing the -SH groups (Varrone 1970). 2-Thenoyl-trifluoroacetone (TTFA) (Gutman and Hartstein 1974) and chlorothricin (Schindler 1975) also inhibit MDH. See also Kun et al. (1967) and Wedding et al. (1967).
Stable for one year when stored at 2 - 8°C as a suspension in ammonium sulfate.
Various organic compounds have been shown to have a significant stabilizing effect (George et al. 1969).