For our international customers, please be advised that orders cannot be placed through our website by customers in countries with International Distributor representation.
Oxalate Decarboxylase - Manual
Oxalate decarboxylase has been isolated from Aspergillus niger ( Emiliani and Bekes 1964) and Myrothecium verrucaria (Lillehoj and Smith 1965). That purified by Shimazono and Hayaishi (1957) from the mycelum of Collybia velutipes, was purified about 400-fold and is reported most useful in the specific determination of oxalic acid. Hallson and Rose (1974) describe a simple, specific assay for urinary oxalate wherein no preliminary treatment of the urine is required. A change in pH is measured. Mayer et al. (1963) have shown the enzyme suitable for urinary oxalate determination. Crawhill and Watts (1961) used it for plasma oxalate content and Haas and Fleischman (1961) for oxalate in beer. Bengtsson (1967) has reported on the enzymatic determination of oxalic acid in plant tissue.
Characteristics of Oxalate Decarboxylase from Collybia velutipes:
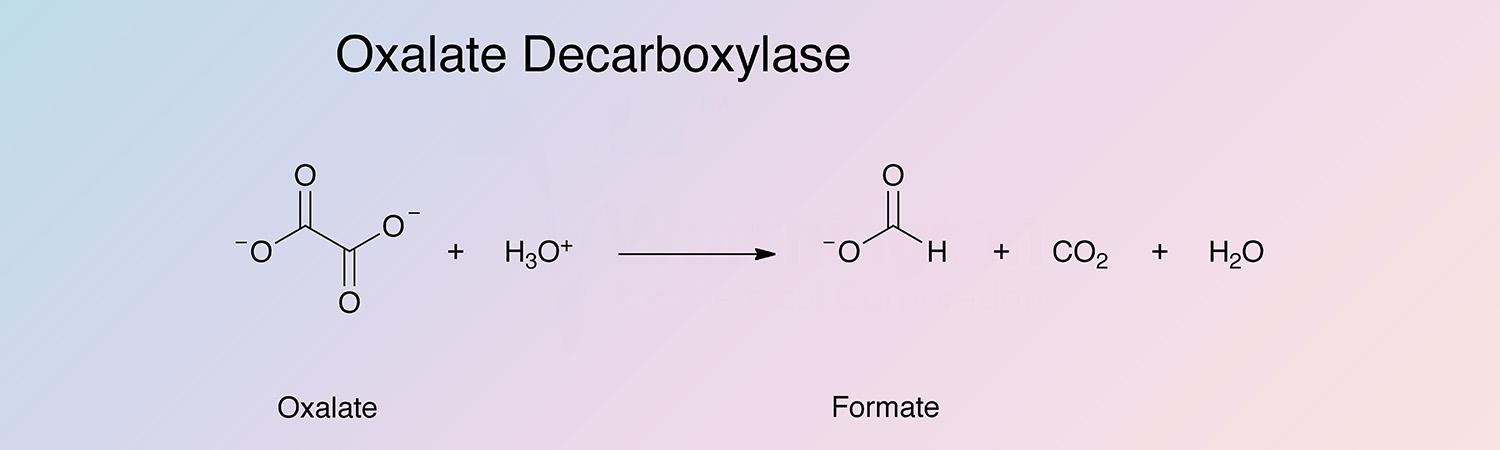
It acts specifically on oxalic acid and produces stoichiometric quantities of CO2 and formic acid. Pyruvate malonate, succinate, glutarate, malate, citrate, oxaloacetate, α-ketomalonate, acetate and formate are not acted upon. (Jakoby 1962).
3.0.