For our international customers, please be advised that orders cannot be placed through our website by customers in countries with International Distributor representation.
Plasma Amine Oxidase - Manual
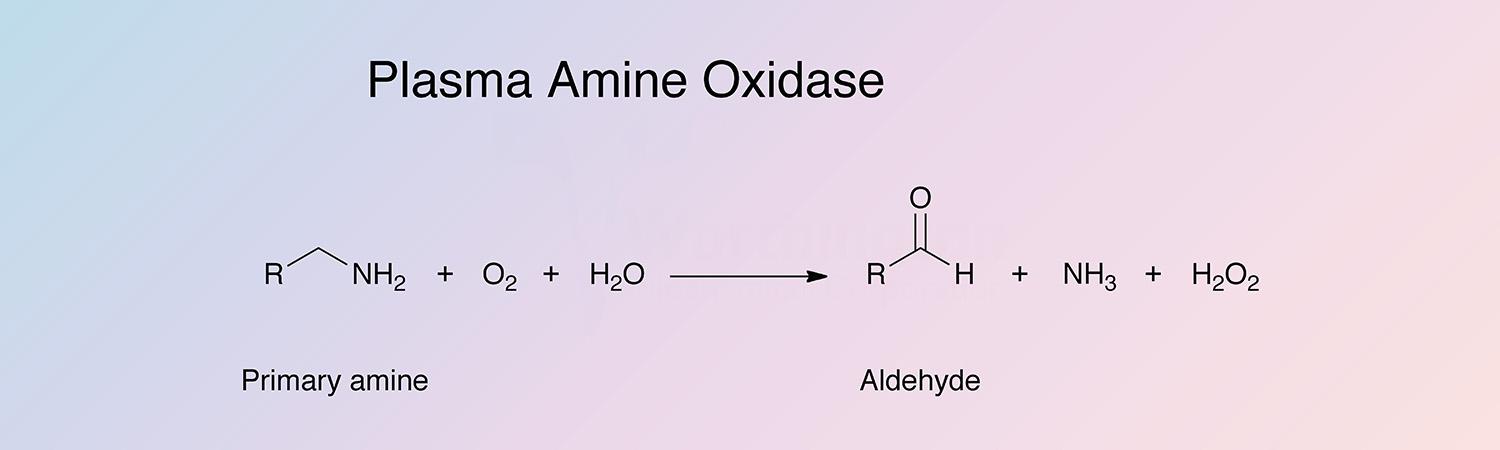
Plasma amine oxidase (PAO) catalyzes the reaction:
There are two classes of amine oxidases: the pyridoxal and copper-containing enzyme to which plasma amine oxidase belongs and the FAD-containing amino oxidases. They play an important biological role. See review by Blaschko (1963). Natural substrates include catecholamines, tryptamine derivatives and other physiologically active amines. Amine oxidases from different sources have different substrate specificities. They have been reviewed by Malmström et al. (1975). Considerable literature exists concerning amine oxidases derived from various organs and animals. The amines spermidine and spermine, most specifically oxidized by bovine plasma oxidase, are significant biochemicals; and have been reviewed by Tabor and Tabor (1972) and by Russell (1973). Russell (1973, 1971; and Russell et al. 1971) report an increased urinary excretion of polyamines by cancer patients. This was further expanded on by Lipton et al. (1975), and by Russell, Durie and Salmon (1975). Analysis of low concentrations of the polyamines can be done by gas chromatography and amino acid analysis (Russell 1973), and electrophoresis (Lipton et al. 1975), but older reports showed utility of plasma amine oxidase for fluorometric (Unemoto et al. 1963) and spectrophotometric analysis (Bachrach and Reches 1966). Similar peroxide-detecting systems Guilbault and Kramer 1964; Gochman and Schmitz 1971) could also be adapted. >Bachrach (1970, 1973) deals with this subject in detail along with many other aspects of polyamine functions.
Characteristics of Plasma Amine Oxidase from Bovine Plasma:
The major physiological amines oxidized are spermine and spermidine (Yamada and Yasunobu 1962; Yasunobu and Smith 1970; Tabor et al. 1954) and some activity is also seen with benzylamine, homosulfanilamide, furfurylamine and simple aliphatic monoamines. (See also Malmström 1975). There is only slight activity with tyramine, and none with tryptamine, epinephrine, serotonin, or agmatine (Yamada and Yasunobu 1962; Tabor et al. 1954).
Achee et al. (1968) indicate that the enzyme is composed of two identical polypeptide chains. There are two pyridoxal phosphates and two atoms of Cu2+ per molecule (Yamada and Yasunobu 1962 and 1963). The amino acid composition is listed by Malmström et al. (1975).
170,000 (Achee et al. 1968).
For spermine, 6.2, for spermidine, 7.2. Other amines may have other pH optima (Tabor et al. 1954).
![]() = 9.8 (Yamada and Yasunobu 1962).
= 9.8 (Yamada and Yasunobu 1962).
For spectra at various pH values, see Yamada and Yasunobu (1963).
Copper chelating agents, many carboxyl reagents such as cuprizone and hydroxylamine; cyanide (Yamada and Yasunobu 1963). Benzoic acid and benzyl alcohol are both non-competitive inhibitors (Ki=30 and 34 mM respecitvely), (Wang et al. 1968). Tabor et al. (1954) give good summary of inhibitors and the paper by Bardsley et al. (1974) dealing with human placental amine oxidases has a large amount of inhibitor data that may be relevant to this bovine plasma amine oxidase also.
Yamada and Yasunobu (1962) report that impure enzyme in 0.03 M phosphate buffer between the pH values of 6.0 to 8.0 is fairly stable when refrigerated, but that highly purified preparations are less stable. Both forms require electrolytes to prevent precipitation of amorphous protein. It was recommended that the enzyme be stored as a suspension in 55% saturated ammonium sulfate in 0.03 M phosphate buffer, pH 7.0, at -5°C to 2°C. Lyophilization causes 15-20% loss of activity, but provides a product of adequate stability (Yamada and Yasunobu 1962).