For our international customers, please be advised that orders cannot be placed through our website by customers in countries with International Distributor representation.
Pyruvate Kinase - Manual
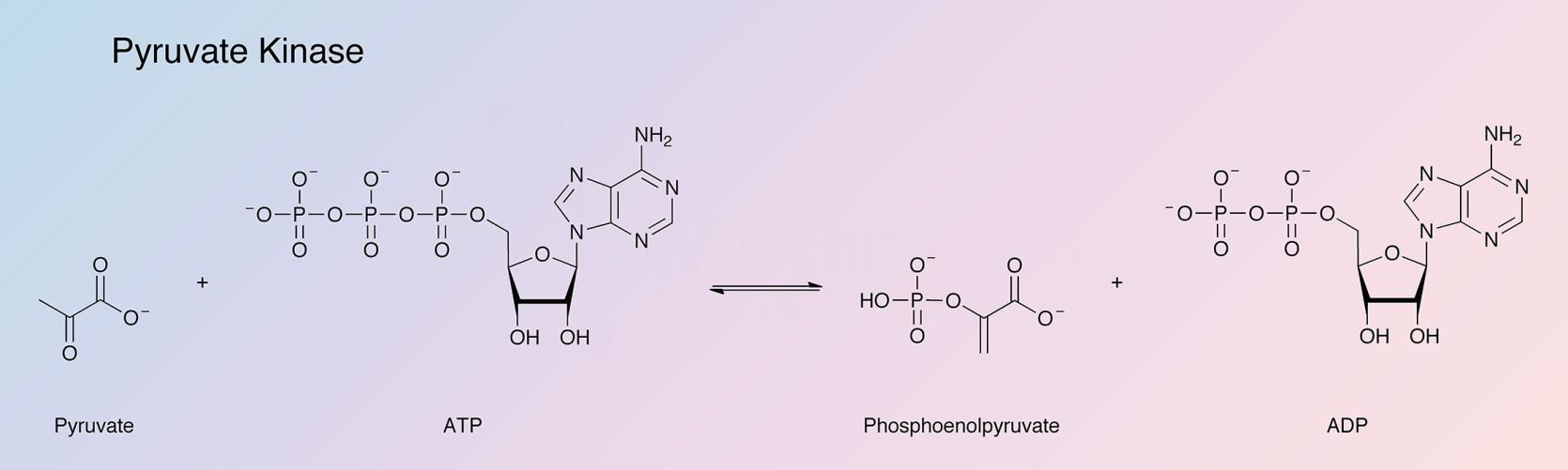
Pyruvate kinase catalyzes the reaction:
Pyruvate kinase (PK) is a key enzyme in glycogen metabolism. Mammalian PK of different tissues are distinct, their characteristics being related to tissue metabolic requirements (Bigley et al. 1974; Boivin and Galand 1974). The enzyme has been well reviewed by Kayne (1973).
The pyruvate kinase most readily obtainable and hence most widely studied, is that from rabbit muscle. It has found extensive use in the quantitative determination of ADP and of enzymes that catalyze the formation of ADP.
Characteristics of Pyruvate Kinase from Rabbit Muscle:
Hohnadel and Cooper (1973) report on specificity with respect to the nucleotide substrate. Phosphate from phosphoenolpyruvate (PEP) is transferred to ADP, GDP, UDP, or CDP with activity decreasing in this order. (See also Kayne 1973.)
The enzyme is tetrameric (subunit M.W.: 57,000) with four metal binding sites (Melamud and Mildvan 1975). On subunit dissociation, a dimeric intermediate is formed (Davies and Kaplan 1972). There is no covalently bound prosthetic group. Cottam et al. (1969) determined the constituent amino acids and McPherson and Rich (1972) an x-ray structural analysis. Kwan et al. (1975) have reported on the metal-binding sites. PK has an absolute requirement for a divalent metal ion and a monovalent metal ion. Mg2+ and K+ probably fill these needs in vivo; (Kayne and Price 1972). Erhard and Davis (1975) indicate that the same active site catalyzes hydrolysis of phosphoenolpyruvate. Flashner et al. (1972 and 1973) indicate that four sulfhydryl groups have a role at the active site. (See also Nowak and Mildvan 1972). Brevet et al. (1975) characterize two binding sites, one for the nucleoside substrate (metal dependent) and one for the acceptor.
237,000 (Cottam et al. 1969).
7.5 (Bücher and Pfeiderer 1955).
![]() = 5.4 (Cottam et al. 1969).
= 5.4 (Cottam et al. 1969).
Activity is inhibited by Ca2+ (Betts et al. 1968; Mildvan and Cohn 1965). Fluorophosphate inhibits the enzyme reaction competitively with respect to PEP (Mildvan and Cohn 1967). ATP inhibits the reaction by removal of Mg2+ from the substrate MgADP-. ATP also appears to inhibit the reaction competitively with respect to both ADP and PEP if the Mg2+ concentration is higher than that of ATP (Holmsen and Storm 1969). Inactivation is also observed in the presence of pyridoxal 5'-phosphate (Johnson and Deal 1970). Creatine phosphate appears to be competitive with PEP (Kemp 1973). Bondar and Pon (1969) reported evidence of an endogenous inhibitor removable by chromatography. See also Davidoff and Carr (1973) and Kayne and Price (1972).
The kinetics of the reaction have been dealt with by Ainsworth and MacFarlane (1973). PK, in addition to its phosphorylation of ADP by phosphoenolpyruvate, also catalyzes the phosphorylation of fluoride (Tietz and Ochoa 1958), hydroxylamine and glycolate (Kayne 1974) by ATP. Anderson and Randall (1975) indicate that rabbit muscle enzyme is not activated by fructose-1,6-diphosphate as in sturgeon muscle and rabbit liver PK. Giles et al. (1975) have reported on the kinetics of the reverse reaction. See also Erhard and Davis (1975).