For our international customers, please be advised that orders cannot be placed through our website by customers in countries with International Distributor representation.
Proteinase K - Manual
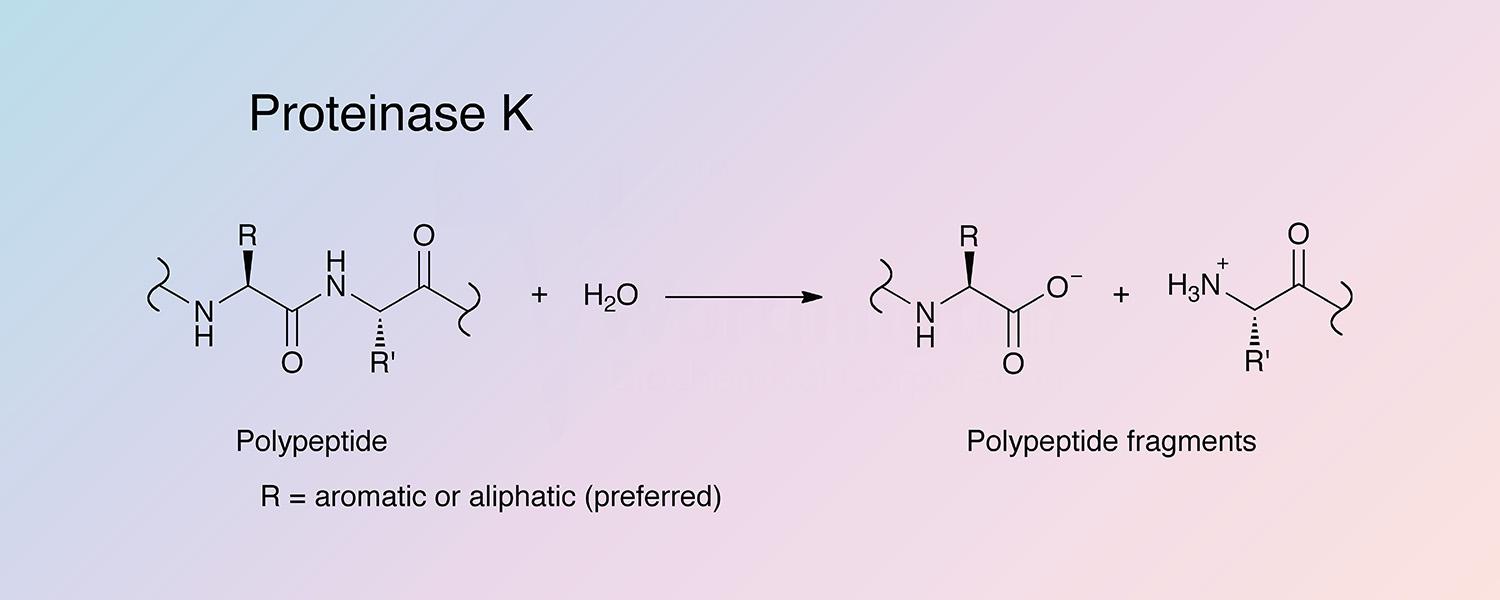
Proteinase K (PROK) is a serine endopeptidase with a broad spectrum of action, isolated from the filtrate of the fungus Tritirachium album limber.
Characteristics of Proteinase K from Tritirachium album limber:
In addition to cleavage of peptide bonds, it is able to catalyze peptide amide hydrolysis. Proteinase K is inactivated by diisopropyl fluorophosphate (DFP) or phenyl methane sulphonyl fluoride (PMSF). Chelating agents such as citrate and EDTA have no affect on the enzyme activity.
Proteinase K is very useful in the isolation of highly native, undamaged DNAs or RNAs, since most microbial or mammalian DNases and RNases are rapidly inactivated by the enzyme, particularly in the presence of 0.5 - 1% SDS.
28,900 daltons.
7.5 - 12, using denatured hemoglobin as substrate.
PROK/PROKR: Store at 2 - 8°C; PROKS/PROKRS: Store @ -20°C
Worthington PROK/PROKR is supplied as a highly purified lyophilized powder. Code PROKS/PROKRS is a 20mg/ml solution containing 50% glycerol. Both are tested to be free of DNase and RNase.
Although calcium ions do not affect the enzyme activity, they do contribute to its stability when present at a concentration of 1 - 5 μmoles. An interesting characteristic of proteinase K is that it retains its activity in the presence of sodium dodecyl sulphate (SDS) or urea. (0.5 - 1% SDS and 1 - 4 M urea). Raising the temperature of the reaction from 37°C to 50°C - 60°C can increase the activity several folds. A special feature of proteinase K is its ability to digest native proteins, thereby inactivating enzymes such as DNase and RNase without recourse to a denaturation process.