For our international customers, please be advised that orders cannot be placed through our website by customers in countries with International Distributor representation.
Phosphodiesterase II - Manual
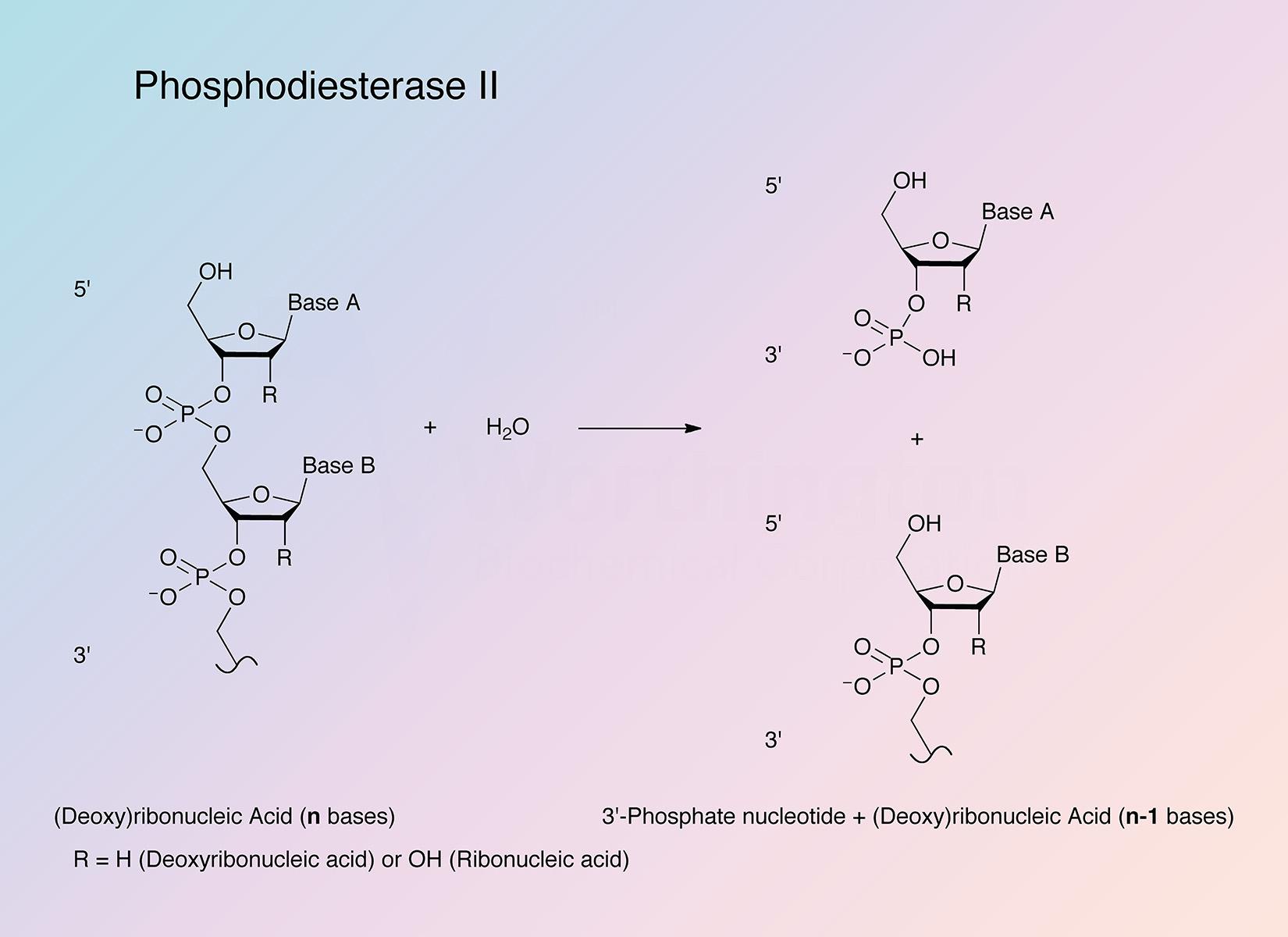
Spleen exonuclease (Phosphodiesterase II) hydrolyses 3'-phosphomononucleotides from oligonucleotides containing a 5'-hydroxyl terminus. See review by Bernardi and Bernardi (1971).
The enzyme's specificity for the 5'-phosphate end of a polynucleotide chain after conversion to 5'-OH by use of E. coli alkaline phosphatase makes spleen exonuclease very important in the characterization of polynucleotide chain length, base composition and identity of terminal nucleotide. See Ho and Hilham (1973), Philippsen and Zachau (1971), Roychoudhury et al. (1971), and >Sneider (1971).
Characteristics of Exonuclease from Bovine Spleen:
Spleen exonuclease splits off, sequentially, nucleoside 3'-phosphates, starting at the 5'-OH end, from ribo- or deoxyribo-oligonucleotides. RNA "core", alkali-treated DNA, poly(A), poly(U), and poly(I) are acted upon. Prior removal of the 5'-terminal phosphate group is essential.
Native DNA and poly(C) are quite resistant. Bis-(p-nitrophenyl) phosphate is a very poor substrate. (Bernardi and Bernardi 1968).
5.5 using succinate and phosphate buffer and an acid DNase digest; pH 6-7 with 0.1 M acetate buffer. (Bernardi and Bernardi 1971).