For our international customers, please be advised that orders cannot be placed through our website by customers in countries with International Distributor representation.
Protease, Staph aureus (Endoproteinase Glu-C) - Manual
Source:
Staphylococcus aureus V8
CAS:
66676-43-5
EC:
3.4.21.19
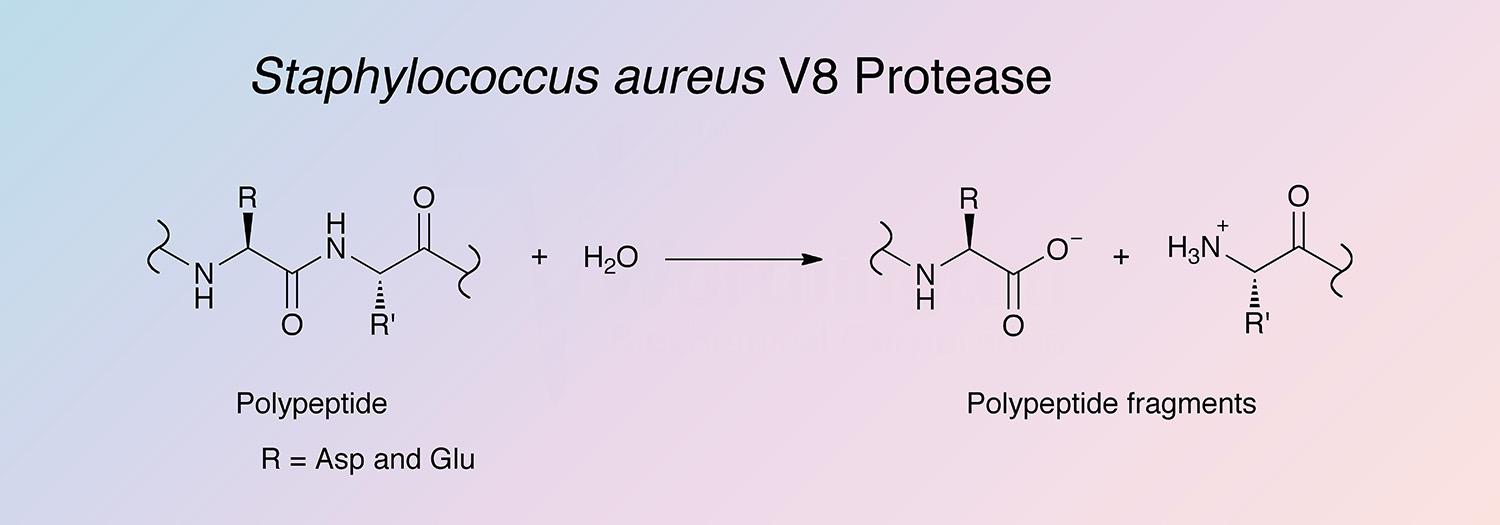
Protease S. aureus, V8 (endoproteinase-Glu-C) specifically cleaves peptide bonds on the COOH-terminal side of either aspartic or glutamic acids (Drapeau et al. 1972). Houmard and Drapeau (1972) report that in the presence of ammonium buffers the enzyme specificity can be limited to glutamoyl bonds. Its rather unique specificity, which can be considered as opposite to that of trypsin, makes it a useful tool for protein chemistry and peptide mapping studies (Cleveland et al. 1977) and (Hall et al. 1978).
Characteristics of Protease from S. aureus, V8:
Characteristics of Protease, Staph aureus (Endoproteinase Glu-C)
Molecular Weight
27,000 (Drapeau 1978).
Optimal pH
4.0 and 7.8 with hemoglobin substrate. (Drapeau et al. 1972).
Extinction Coefficient
![]() = 4.26 (Houmard 1976).
= 4.26 (Houmard 1976).
Inhibitors
Diisopropyl fluorophosphate (DFP) and monovalent anions such as F-, Cl-, Br-, CH3COO-, and NO3- (Houmard 1976).
Protease, Staph aureus (Endoproteinase Glu-C) Products
Description
Activity
Code
Cat. #
Size
Price
Description
Protease, S. aureus, Sequencing Grade
Source:
Staphylococcus aureus V8
Chromatographically purified according to Drapeau, G, et.al, JBC, 247, 6720 (1972). Supplied as a lyophilized powder.
Store at 2-8°C.
Code
STSEQ
Product details
LS02126
5x10 ug
$200.00
LS02128
5x50 ug
$540.00
LS02129
Bulk
---
Description
Protease, S. Aureus (Endoproteinase Glu-C)
Source:
Staphylococcus aureus V8
Chromatographically purified according to Drapeau, G., Boily, Y., and Houmard, J.: JBC, 247, 6720 (1972). A lyophilized powder.
Store at 2-8°C.
Code
STAP
Product details
LS003608
1 mg
$79.00
LS003605
5 mg
$280.00
LS003606
Bulk
---