For our international customers, please be advised that orders cannot be placed through our website by customers in countries with International Distributor representation.
Hyaluronic Acid - Manual
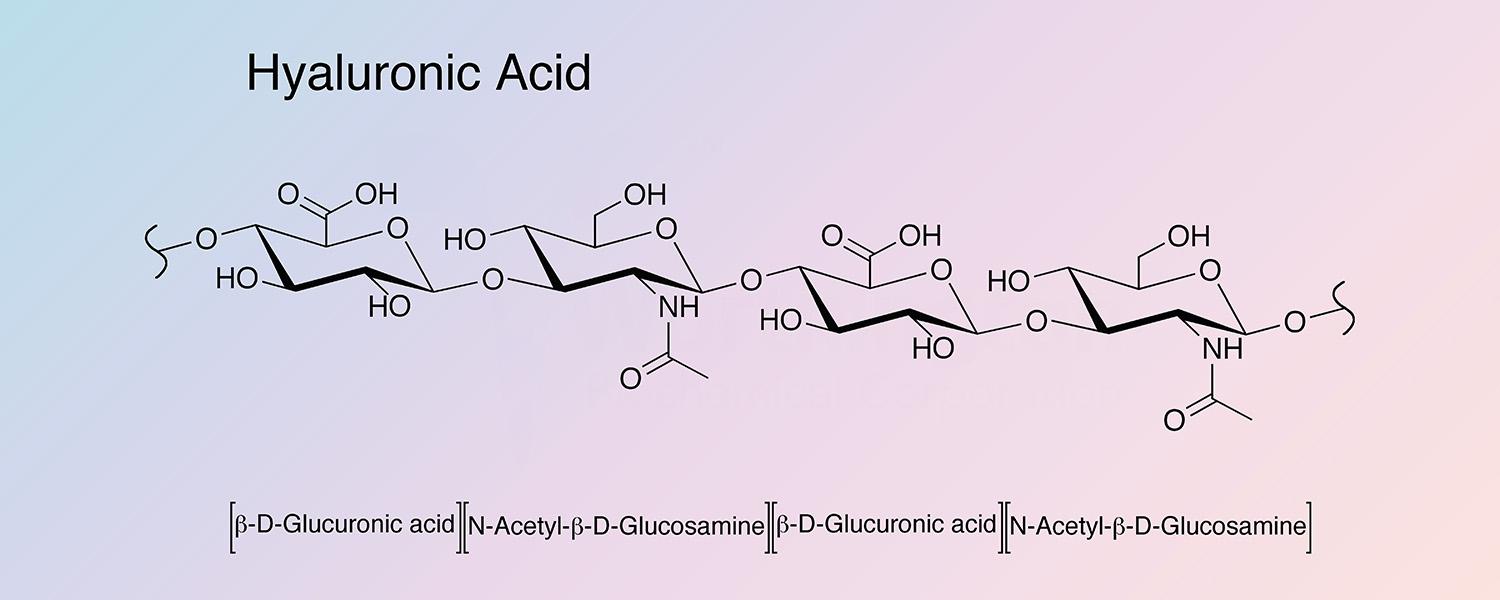
The hyaluronic acids (HA) are a class of macromolecular proteoglycans characterized by a highly polymerized chain of glucuronic acid and N-acetylglucosamine units bonded to protein. They exist in nature as a hydrated gel, usually closely associated with other tissue components such as chrondroitin sulfate. (Arkins and Sheehan 1972 and Bettelheim and Philpott 1959).
Occurring in intercellular ground substance of connecting tissue (Blumberg et al. 1955), they have an important role in controlling tissue permeation, bacterial invasiveness and macromolecular transport between cells (Laurent et al. 1975). HA is also one of the proteoglycans found in cartilage (Hardingham and Muir 1973). It has been indicated that hyaluronic acid is important in controlling tissue hydration (Block and Bettelheim 1970). In synovial fluid and heart valve its purpose would appear to be primarily that of a lubricant (Meyer et al. 1969). HA is present in the vitreous humor of the eye and fluids of the inner ear. Its ability to act as a mechanoelectric transducer makes possible intriguing speculation in regard to its function (Barrett 1975).
The ratio of glucuronic acid to glucosamine varies with the source as does the protein or peptide residue remaining after exhaustive protease digestion (Meyer et al. 1969; Silpananta et al. 1968; Taussig 1965; Marcante 1965; Pigman 1961; and Preston et al. 1965).
Characteristics of Hyaluronic Acid from Bovine Vitreous Humor:
The molecular weight of HA preparations varies with purification procedures, i.e., the extent of degradation as well as the source. According to Bettelheim (1958) when the protein is completely removed a molecular weight on the order of 70,000 is obtainable as compared with 2-4 million in a highly polymerized preparation (Laurent and Gergely 1955). It has been reported (Laurent et al. 1960) that bovine vitreous humor hyaluronic acid has a lower molecular weight than most other sources and that from human umbilical cord has the largest molecular weight (Jensen and Carlsen 1954). Pigman et al. (1961) report that HA is degraded in the presence of oxygen and reducing agents and by merthiolate, often used in its preparation. Ascorbic acid likewise has this effect (Swann 1967). The carbohydrate polymer is negatively charged. When it is mixed with a cationic protein such as albumin at low pH, a precipitate is formed. If the glycosidic bonds have been split, i.e., the chain depolymerized, this precipitation no longer occurs. This phenomenon is the basis for the turbidimetric assay of hyaluronidase.