For our international customers, please be advised that orders cannot be placed through our website by customers in countries with International Distributor representation.
Glucose-6-Phosphate Dehydrogenase - Manual
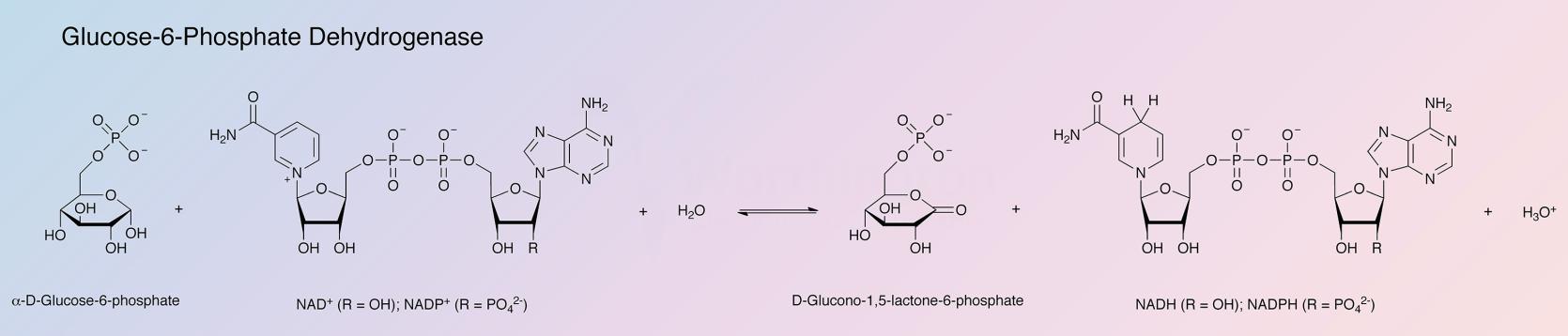
Glucose-6-phosphate dehydrogenase (G6PD) is a regulatory enzyme catalyzing the first step of the pentose phosphate pathway: oxidation of glucose-6-phosphate using NADP+ and/or NAD+.
![]()
The G6PD of L. mesenteroides has been studied in great detail because of its unique dual-coenzyme specificity. Most dehydrogenases prefer either NAD+ or NADP+, but L. mesenteroides G6PD can use either.
The bacterial genus Leuconostoc first gained interest in the late 1920s and early 1930s as a means of studying lactic fermentation. Pederson determined that glucose fermentation yields equal quantities of lactate, ethanol, and CO2, which was later confirmed by Friedemann (Pederson 1929, and Friedemann 1939).
In 1951, De Moss et al. first isolated glucose-6-phosphate dehydrogenase from L. mesenteroides. De Moss et al. also demonstrated its dual nucleotide specificity in 1953, and in 1955 showed that fermentation by L. mesenteroides proceeds via a mechanism differing from the classical Embden-Meyerhof glycolytic scheme.
In the 1960s, Kemp and Rose demonstrated that the two reduced coenzyme products have different metabolic roles in vivo: NADPH is used in biosynthetic reactions and NADH is used in ATP-generating reactions in the production of ethanol and lactate (Kemp and Rose 1964, and White and Levy 1987).
In the late 1980s and into the 1990s, critical amino acid residues were identified, and it was shown that the kinetic mechanisms of NAD- and NADP-linked reactions differ (Levy 1989). In 1991, Lee et al. published the complete amino acid sequence of the monomer.
Current research has shown that in the case of G6PD from L. mesenteroides the enzyme is active as both a homodimer and monomer, whereas the enzyme from yeast is active only in the native, homodimeric form (Ravera et al. 2010). G6PD has also been coupled to other enzymatic reactions, where it functions to regenerate coenzyme (NADH) (Ohno et al. 2008). The stability of the enzyme and its resistance to inactivation while bound to substrates also has recently been investigated (Duggleby 2007).
Either NAD or NADP will serve as coenzyme with the intrinsic reaction velocity of NAD being approximately 1.8 times greater than that of NADP (Olive and Levy 1967). D-glucose-6-phosphate is considered to be the natural substrate, although D-glucose reacts slowly (Metzger et al. 1972).
Asp177 and His240 form the catalytic dyad of G6PD. His240 acts as a base to extract the C-1 proton from glucose-6-phosphate; Asp177 stabilizes the resulting positive charge (Cosgrove et al. 1998).
L. mesenteroides G6PD is a dimer of identical subunits with two active sites per dimer putatively located in a pocket between the coenzyme binding site and the large beta+alpha domain in each subunit (Rowland et al. 1994). This pocket contains several completely conserved amino acids including an eight residue peptide (Cosgrove et al. 1998).
The L. mesenteroides G6PD gene encodes a polypeptide of 485 amino acids. G6PD is unique in that it contains no cysteine residues. The sequences of human and E. coli, which use only NADP+ under physiological conditions, have been determined. The sequence of Z. mobili, which like L. mesenteroides can use either NADP+ or NAD+, has also been determined. There is approximately 30-36% sequence identity between any two of these species. A conserved sequence begins at Arg175, and contains a lysine residue thought to function in glucose-6-phosphate binding (Lee et al. 1991).
- Systems where NADPH or NADH is measured (Bhattacharya and Ali 1988)
- Determination of glucose and ATP (when coupled with HK), fructose, glucose-6-phosphate, NAD(P)+, and CK
P11411
Two Domains:
- Class: Alpha Beta and Alpha Beta
- Architecture: 3-Layer(aba) Sandwich and 2-Layer Sandwich
- Topology: Rossmann fold and Dihydrodipicolinate Reductase (domain 2)
Molecular Weight:
- 108.6 kDa (Theoretical)
- 7.8, Tris-HCl buffer, 0.05 M and NAD as coenzyme
- 4.6 (Olive and Levy 1971)
- 133,420 cm-1M-1 (Theoretical)
- E1%, 280.5 = 11.5, pH 7.2 in 0.1 M Tris-HCl (Olive and Levy 1971)
- Aspartic acid (D177)
- Histidine (H240)
- Stabilized by BSA
- Modestly stimulated by HCO3- (≤ 0.3 M)
- Acetyl-CoA (Olive and Levy 1975)
- High concentrations of palmitoyl-CoA
- ATP (Olive and Levy 1975)
- CoA (Olive and Levy 1975)
- 1-Fluoro-2,4-dinitrobenzene (irreversible)
- Pyridoxal 5’-phosphate with respect to ZF (competitively) and NAD or NADP (noncompetitive)
- L. mesenteroides G6PD, unlike mammalian G6PDs, is unaffected by steroids (Lee et al. 1991)