For our international customers, please be advised that orders cannot be placed through our website by customers in countries with International Distributor representation.
Collagenase - Manual
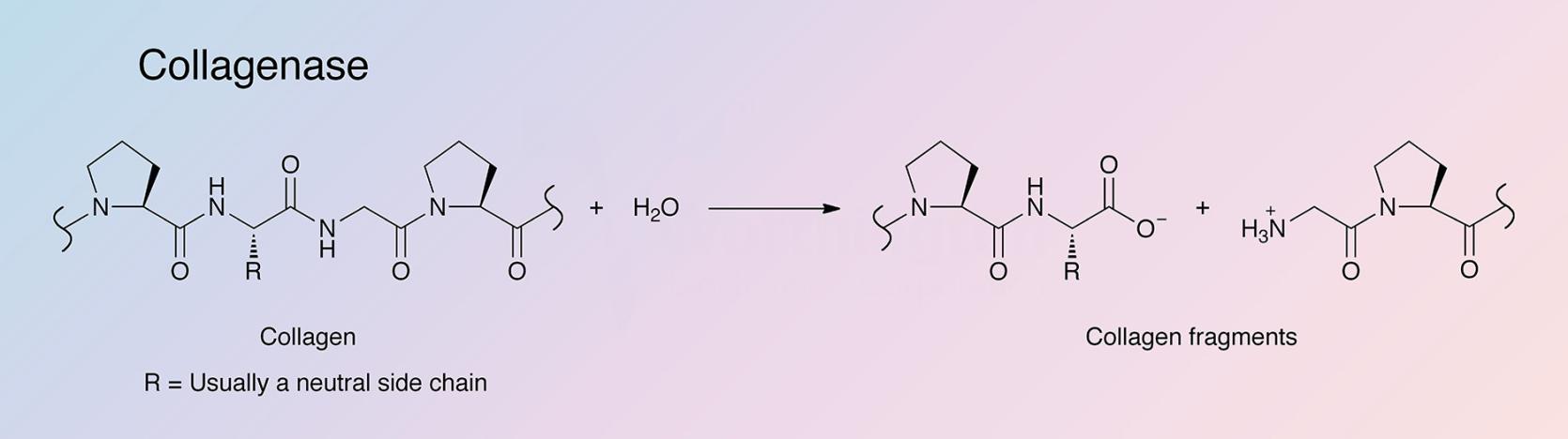
Collagenases are endopeptidases that digest native collagen in the triple helix region. Collagens are the major fibrous component of animal extracellular connective tissue. Bacterial collagenases differ from vertebrate collagenases in that they exhibit broader substrate specificity (Peterkofsky 1982, Birkedal-Hansen 1987). Unlike animal collagenases that split collagen in its native triple-helical conformation (Woolley et al. 1975, Gross et al. 1974), bacterial collagenase is unique because it can degrade both water-insoluble native collagens and water-soluble denatured ones. It can attack almost all collagen types, and is able to make multiple cleavages within triple helical regions (Mookhtiar and Van Wart 1992).
Worthington Collagenases:
• Purified collagenase, Codes: CLSPA/CLSPANK, contain minimal secondary proteolytic activities along with high collagenase activity.
• Type 1 partially purified collagenase has the original balance of collagenase, caseinase, clostripain and tryptic activities.
• Type 2 contains higher relative levels of protease activity, particularly clostripain.
• Type 3 contains lowest levels of secondary proteases.
• Type 4 is designed to be especially low in tryptic activity to limit damage to membrane proteins and receptors.
• Type 5 contains higher collagenase and caseinase values.
• Type 6 contains high collagenase activity with a caseinase to collagenase ratio ~2:1. Designed to be enriched for Type II (col H) collagenase relative to Type I (col G).
• Type 7 contains collagenase and caseinase activities four-fold higher than collagenase Types 1 and 2.
Animal Free Types AFA, AFB, AFC, STZ1 and STZ2 collagenases are derived from cultures grown in medium completely devoid of animal based components and designed for bioprocessing applications where introduction
of potential animal derived pathogens must be prevented. Levels of secondary proteases are similar to Types 1 and 2 collagenase.
- CLSAFA is the original AF grade designed to have collagenase and secondary proteases similar to Types 1 and 2 collagenase.
- CLSAFB contains higher collagenase and caseinase activities than CLSAFA.
- CLSAFC has especially low tryptic activity similar to Type 4 collagenase.
- CLSAFD contains two to three fold higher specific activity than CLSAFA
- Purified collagenase , code CLSAFP contains minimal secondary proteolytic activities along with high collagenase activity
- • STZ1 & STZ2, 0.22μ filtered STEMxyme® AF Collagenase/Neutral Protease (Dispase®) blends for primary and stem cell isolation.
Crude preparations contain not only several collagenases but also a sulfhydryl protease, clostripain (Mitchell 1968), a trypsin-like enzyme (Peterkofsky and Diegelmann 1971, Sparrow and McQuade 1973), and an aminopeptidase (Kessler and Yaron 1973). Sugasawara and Harper (1984) and Bond and Van Wart (1984) report on purification of the collagenases of Cl. histolyticum.
Much of what is presently known about the characteristics of clostridial collagenase comes from the pioneering studies in the 1950s by Mandl, Seifter, Harper and their associates, and the later classification work of Van Wart and Bond (Mandl et al. 1953, Mandl et al. 1958, Seifter et al. 1959, Harper et al. 1965, Bond and Van Wart 1984).
In 1959, the first commercially available collagenase isolated from Clostridium histolyticum was offered by Worthington. At that time, only one preparation of crude enzyme was offered.
After collagenases from Clostridium histolyticum were first prepared by Mandl et al. (1953), studies from the late 1950s to the mid 1980s found that several separable collagenases exist and these fractions’ specificities and stabilities were partially characterized. (Grant and Alburn 1959, Mandl et al. 1964, Yoshida and Noda 1965, Kono 1968, Seifter and Harper 1970, Harper and Kang 1970, Lwebuga-Mukusa et al. 1976, Bond and Van Wart 1984). The molecular weights of these seven collagenases were found to range from 68 kDa to 130 kDa, and were classified as either Class I or Class II, based on a variety of properties (Bond and Van Wart 1984). These classes were found to differ with respect to their activities, stabilities, and amino acid composition, but they share many similarities.
Until 1962, most interest in collagenases centered on clostridial collagenases. In that year, Gross and Lapiere obtained evidence for a collagenase in bullfrog tadpole tissue culture media (Gross 1962, Harper 1980), which was the discovery of the first vertebrate collagenase. Subsequent to this discovery, a large number of collagenases were found in marine life, other bacteria (Schoellmann and Fisher 1966, Welton and Woods 1973 and 1975, Keil et al. 1975), amphibians, and mammals.
Further studies of collagenases from human and other mammalian sources were reported on, and continue to be actively studied in order to better understand the pathology and treatment of human diseases. Of particular interest is the relationship between collagenase and rheumatoid arthritis (Abe and Nagai, 1973, Kruze and Wojtecka 1972, Bauer et al. 1971, Evanson et al. 1968), metastasis (Harris et al. 1972, McCroskery et al. 1973 and 1975, Alaho et al. 2005), wound debriding (Hoover 1959), herniated disc treatment (Sussman 1968), angiogenesis, tissue repair, and cirrhosis.
The hyper-reactive sites where the Class I and Class II enzymes initially attack all three collagen types were identified by French et al. (1992). The cleavage sites are all at Yaa-Gly bonds in the repeating Gly-X-Y collagen sequence (Barrett 1998). Clostridial collagenases’ ability to digest native, triple-helical types I, II, and III collagens into a mixture of small peptides is its primary distinguishing factor. This is accomplished by making multiple scissions in the triple helix. Digestion is completed by hydrolyzing those fragments into a mixture of small peptides. Conversely, vertebrate collagenases initiate collagenolysis by making a single scission across all three alpha chains after which attack on those alpha chains is very limited. Gelatinases and other proteases then carry out collagenolysis only after denaturing of the triple helix (Barrett 1998).
Clostridium collagenases represent unusually large metalloproteases, a family of proteases that shares a zinc-containing motif at the center of the active site (Gonzales and Robert-Baudouy 1996).
Collagenase is produced by two separate and distinct genes in Clostridium histolyticum. Both genes have been cloned and sequenced (Yoshihara 1994). The colG gene codes for type I collagenase, a 936 amino acid peptide. The colH gene codes for type II collagenase, a 1021 amino acid peptide. These genes share 72% identity, and the proteins share 43% identity. Both gene products can be present as two or more isoforms differing in molecular weight. Crude collagenase mixtures can therefore contain six to eight different molecular weight species ranging from 68 to 130 kDa. Substrate specificity studies have demonstrated that the colG gene prefers natural substrates such as intact collagen, compared to the colH gene product. Conversely, the colH gene product preferentially acts on short synthetic substrates (FALGPA) relative to the colG gene product (Eckhard et al. 2009 and Matsushita 1999).
- Isolation of adipocytes, hepatocytes, and cells from lung, epithelium and adrenal tissue
- Isolation of cardiomyocytes and cells from bone, cartilage, muscle, thyroid, and endothelium
- Isolation of mammary and various other soft tissues
- Isolation of human and porcine pancreatic islet cells (Kin 2007)
- Treatment of tissues with crude collagenase, with its mixture of proteolytic activities, provides gentle, selective digestion of the intercellular matrix with little damage to cells or loss of viability
- AFA collagenase is suitable for applications needing to avoid introduction of animal derived pathogens into bioprocessing procedures
For collagen structural and biosynthetic studies researchers generally use more highly purified collagenase preparations free of other proteolytic activities.
For tissue dissociation most researchers employ either crude collagenase preparations, such as CLS1, CLS2, CLS3 and CLSAFA combined with other enzymes such as elastase, trypsin, and/or papain. For more information, please see the Collagenase Sampling Program.
- Q9X721 (ColG, Theoretical)
- Q46085 (ColH, Theoretical)
- Class: Mainly beta
- Architecture: Sandwich
- Topology: Jelly rolls
68-130 kDa
6.3-8.5 (Soru and Zaharia 1972)
- 5.63 (ColG, Theoretical)
- 5.58 (ColH, Theoretical)
- 159,970 cm-1 M-1 (ColG, Theoretical)
- 150,130 cm-1 M-1 (ColH, Theoretical)
- E1%,280 = 13.20 (ColG, Theoretical)
- E1%,280 = 13.40 (ColH, Theoretical)
- Ca2+
- Zn2+
- EDTA, EGTA
- Cysteine, histidine
- DTT
- 2-mercaptoethanol
- o-phenanthroline
- Hg2+, Pb2+, Cd2+, Cu2+
- Not inhibited by DFP or serum
(White and White 1997)
Collagenase is also inhibited by α2-macroglobulin, a large plasma glycoprotein (Werb et al. 1974). Nagase et al. (1983) and Stricklin and Welgus (1983) report on natural collagenase inhibitors. The human skin enzyme is inhibited by human serum (Eisen et al. 1970), but granulocyte collagenase is not (Lazarus et al. 1968). Human serum contains α2-macroglobulin and α1-antitrypsin that may inhibit certain collagenases as well as a third inhibitor reported by Woolley et al. (1975). It has also been shown that collagenase A is photo-inactivated in the presence of methylene blue (Takahashi and Seifter 1970 and Karakiulakis 1991).
Collagenase Products
(col H) collagenase.