For our international customers, please be advised that orders cannot be placed through our website by customers in countries with International Distributor representation.
Worthington Biochemical Technical FAQ
Tissue Dissociation
Answer
What is an acceptable enzyme diluent for Collagenase (CLS)?Collagenase may be directly dissolved in a balanced salt solution or buffer of choice. It should be sterile filtered through a 0.22um pore size membrane. Its optimum pH range is 7-9.
Answer
Can the Neonatal Cardiomyocyte Isolation System (NCIS Kit) be used on adults?
The NCIS kit is specifically packaged for five separate tissue dissociations, each containing up to twelve neonatal rat hearts. The NCIS protocol is available online here. Adult rat cardiomyocytes usually require perfusion ie. Langendorf Perfusion System and Collagenase Type II.
Answer
What are the volumes of Hank's, Earle's and Leibovitz L-15 Media that you sell?
Sterile calcium and magnesium free Hank's Balanced Salt Solution, p 7.4 is supplied in the NCIS (Neonatal Cardiomyocyte Isolation System) kit. A 500ml bottle may be purchased, catalog #LK003210.
The NCIS kit also contains a pouch of Leibovitz L-15 Media Powder. Reconstitute the entire contents of the pouch into a beaker containing 800ml of cell culture grade water. Rinse pouce 2-3 times with additional 100ml. Bring total volume to 1 liter and filter through a 0.22 micron pore size filter. The pouch containing Leibovitz L-15 Media powder for 1 liter is catalog #LK003250.
Sterile Earle's Balanced Salt Solution (EBSS) with calcium, magnesium, bicarbonate and phenol red is supplied with the PDS (Papain Dissociation System) kit. A 100 ml bottle may be purchased, catalog #LK003182.
Answer
What is an acceptable enzyme diluent for Neutral Protease/Dispase (code NPRO/NPRO2)?
Neutral protease is usually dissolved in neutral balanced salt solution of choice (ie PBS, Hanks, Earles etc). The composition of several balanced salt solutions are posted in our Tissue Dissociation Guide at http://www.worthington-biochem.com/tissuedissociation/working.html
Usual working solutions for Neutral Protease are 0.5-3.0 u/ml but this varies widely and is dependent on application, diluent, incubation time and temperature. We generally recommend diluting the enzyme as needed as the lyophilized powder is stable at 2-8C. Reconstituted enzymes should not be stored at 2 - 8°C. If necessary they can be aliquoted and frozen at -20°C. Avoid repeated freeze-thaw cycles.
Answer
What are acceptable enzyme diluents for Deoxyribonuclease (D/DPFF/DPRF)?
D/DPFF: May be dissolved in 5mM acetate, 1 mM calcium, pH 4.5 and stored in single use aliquots at -20 or -70 for 1 yr.
DPRF: Use water or any buffer pH 4-9 except phosphate, avoid calcium chelators, add 50% glycerol for storage as liquid at -20C. Aliquot avoid repeated freeze/thaw cycles.
Special care must be taken with deoxyribonuclease. Mix gently to avoid shear denaturation. In general, Dnase I is activated by bivalent metals. Maximum activation is attained with Mg+ and Ca+. Dnase I is inhibited by EDTA and SDS. The optimum pH of Deoxyribonuclease I from Bovine Pancreas is 7.8.
Answer
What is an acceptable enzyme diluent for Elastase (ES)?
The choice of buffer, concentration and pH are usually dependent on application. Elastase is relatively hydrophobic so solubility at concentrations greater than 0.25% and neutral pH are challenging.
Elastase is stable in the pH range 4-10.4. The optimium pH is near 8.5. For cell culture applications where the material is dissolved in neutral balanced salt solutions and higher concentrations (>2.5%) are desired, increasing the pH to 9-10 aids in solubility and then pH can be lowered to neutral to keep cells happy.
Most labs are dissolving directly in balanced salt solution of choice. It is always best to make a fresh solution as needed but if you must store stocks you can aliquot and freeze at -20C or -80C. You would have to determine frozen stability with your conditions. Elastase should be sterile filtered through a 0.22um pore size membrane before use.
Answer
What is an acceptable enzyme diluent for Papain (PAP/PAPL)?
A common working solution of papain is 20 u/ml papain in 1mM L-cysteine with 0.5mM EDTA dissolved in balanced salt solution of choice (ie EBSS etc). EDTA, cysteine and dimercaptoethanol are considered stabilizers. Optimum pH range is 6.0-7.0.
Answer
What is an acceptable enzyme diluent for Trypsin (TRL)?
Stock solutions of trypsin should be made initially by reconstituting in 0.001N HCL. This solution can be diluted in the digestion medium or buffer immediately prior to use. The optimum pH of Trypsin is approximately 8.0.
Answer
What are conversion factors for units of Collagenase (CLS)?
Unfortunately there is no consistent conversion unit between Wunsch and Mandl units.
Worthington assay is based on modified Mandl unit approx. 0.15 Wunsch units equal 200 Mandl units.
Answer
What are conversion factors for units of Deoxyribonuclease (D/DP/DPRFF)?
Note: Kunitz units as reported by other suppliers can be 2-4 times higher than Kunitz units as measured at Worthington. As measured at Worthington one Kunitz unit digests 1 ug of calf thymus DNA in 10 min at 37C in 50mM Tris, 1 mMMg++, 1 mMCa++, pH 7.8. Correlation of digestion units with Kunitz units is different for other DNA and buffer systems. When protease is present, units can not be correlated.
Answer
What are conversion factors for units of Trypsin (TRL)?
1 mg Worthington TRL> 180 TAME units >10,350 BAEE units >3,450 USP/NF units
Answer
What is the enzyme digestion scale?
Image
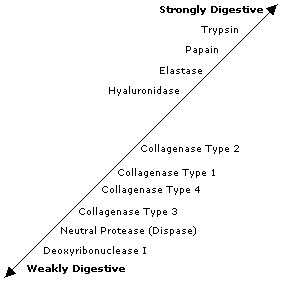
The above scale shows the relative digestive power of the enzymes commonly used for tissue dissociation.
Answer
What are the basic differences among the four types of collagenase?
CLS-1
Contains average amounts of assayed activities (collagenase, caseinase, tryptic and clostripain). Recommended for epithelial, liver (hepatocytes), lung, fat (adipocytes) and adrenal cell isolations.
CLS-2
Contains greater proteolytic activities, especially clostripain activities. It is generally used for heart (cardiomyocytes), bone, muscle, thyroid, cartilage and endothelial cells.
CLS-3
All activities lower than average. Low caseinase and low proteolytic activity. It is usually used for mammary cells and other soft tissues.
CLS-4
Selected because of low tryptic activity. High collagenase required. It is commonly used for pancreatic islets and other applications where receptor integrity is crucial.
Answer
What are the test specifications for each type of collagenase?
collagenase caseinase clostripain tryptic A280 CLS 1
≥125 u/mg
≥200 u/mg
≤4.0 u/mg
≤0.5 u/mg
report
CLS 2
≥125 u/mg
≥200 u/mg
≥3.5 u/mg
≥0.1 u/mg
report
CLS 3
≥100 u/mg
≥50 u/mg
≤3.0 u/mg
≤0.3 u/mg
report
CLS 4
≥160 u/mg
≥100 u/mg
≤3.0 u/mg
≤0.1 u/mg
report
CLSPA
≥500 u/mg
≤50 u/mg
≤2.0 u/mg
≤0.25 u/mg
report
Answer
Does Collagenase Type I include Brain Heart infusion (BHI)?
Brain Heart Infusion (BHI) fermentation media has not used in the the production of Worthington CLS-1 or any Worthington collagenase product.
Answer
What types of media work best with Collagenase I? A colleague of mine uses DMEM but we always use PBS with either 2% FBS or 0.5% BSA. Do you have any suggestions of what might be the best media? We are using it for mouse bone marrow isolation.
Collagenase works well in most balanced salt solutions (ie. Hanks, Earles, PBS, DMEM). The choice is usually more application or tissue specific. We are not working with murine bone marrow here, so I hesitate to choose one over the other. Suggestions are usually based on references. You can search our Tissue Dissociation Guide at www.tissuedissociation.com for references by tissue type.
Answer
What is the expiration time for the neonatal cardiomyocyte isolation system?
The NCIS kits are given one year dating from initial release/assay. You can always request information on reassay date prior to purchase.
Answer
Previously I've used Dispase II at around 2.5 mg/ml at 37C for 2hr or overnight at 4C to dissociate epidermis from dermis in skin biopsies. What concentration of your neutral protease do you recommend for this procedure?
Dispase II usually refers to a partially purified grade of dispase
similiar to Worthington's NPRO2. Historically most lots of NPRO2
average around 0.4 u/mg so 1u/ml of our neutral protease should be
similar to 2.5 mg Dispase II. Neutral protease working concentrations
are usually in the 0.5-2.0 u/ml range.
Answer
Can I use the Neonatal Cardiomyocyte Isolation System with mice?
The Neonatal Cardiomyocyte Isolation System is specifically packaged and use tested on neonatal rats.
A few researchers have used the system on neonatal mice, with some modifications. I have copied some references below for neonatal mice. It seems neonatal mouse hearts may require higher enzyme concentrations and/or longer digestion times than the rats. Increasing the collagenase digestion time to 45 min is a good place to start your optimization. Also one researcher has informed me he increased the trypsin concentration to 100 ug/ml by cutting final volumes in half.
http://ajpcell.physiology.org/cgi/content/full/277/6/C1277#SEC1
http://www.jbc.org/cgi/content/full/277/50/48282
http://www.pnas.org/cgi/content/full/95/16/9669#MaterialsMethods
The protocol for the NCIS kit can viewed by clicking here.
Answer
How can I convert units/mg dw of hyaluronidase into U/ml?
Worthington supplies hyaluronidase as a lyophilized powder reporting the u/mg dry weight on the Certificate of Analysis. You can resuspend hyaluronidase to the desired u/ml using this information.
As an example, the current lot of hyaluronidase was released at 525 u/mg dw, if you need a 500 u/ml solution you would dilute 5 mg with 5.25 ml to obtain the 500 u/ml. Calculation 5mg*525u/mg=2625 units/5.25 ml=500 u/ml
Answer
How do the different types of collagenase offered by Worthington compare to collagenase from other suppliers?
Worthington Collagenase Cross Reference Chart
Worthington CLS-1 Worthington CLS-2 Worthington CLS-3 Worthington CLS-4
Invitrogen/Gibco I
Invitrogen/Gibco II
Invitrogen/Gibco III
Invitrogen/Gibco IV
Roche Collagenase A
Roche Collagenase B
Roche Collagenase D
Sigma Type I
USB type I
Answer
Are your collagenase products tested for endotoxin?
Worthington has instituted endotoxin testing of collagenase. The Limulus amebocyte lysate (LAL) test is used for the quantitation of endotoxin levels in samples. Results are reported on Certificate of Analysis.
Answer
I ordered Collagenase IV from your company (100mg). Some of the protocols call for 2mg/ml concentration and others call for 0.2% solution. How does one prepare a 0.2% solution?
A 0.2% solution is equivalent to 2 mg/ml. Just remember a 1% solution refers to 1 gram per 100 ml or 10 mg/ml.
Answer
Which type of collagenase is present in your purified collagenase, CLSPA?
CLSPA is bacterial collagenase from clostridium histolyticum that is further purified over a column and pooled based on activity. CLSPA has higher collagenase activity (>500 u/mg DW but usually around 1000 u/mg) and significantly lower caseinase, clostripain and tryptic activity than our cruder CLS types 1-4. We do not characterize the specific composition of each lot but it would be a mixture of several collagenases containing both collagenase A and B (sometimes referred to as I and II). A daily listing of Collagenase activities including CLSPA is posted here.
Answer
We ordered hyaluronidase from worthington cat no LS005474. the activity says that it is >/3000 USP/NF units per mg dry weight. Can you please convert it to mg. thanks
Hyaluronidase is sold on a unit basis. The mg supplied may vary slightly between lots as determined by the u/mg activity of each lot. The current lot of HSEP LS005474 is 58E10498 which was released at 7,654 u/mg dry weight. For this lot the 30,000 unit package will contain ~4 mg (30,000 units/7654 u/mg=4mg). If you have a different lot number of HSEP you can check the Certificate of Analysis from the following link- http://www.worthington-biochem.com/index/technical.html
Answer
I have a question relating to your "Neonatal Cardiomyocyte Isolation System" protocol. Is there any serum added to your L-15 media provided in the kit or do I need to add some separately? If the serum is already present, would it be possible to culture the cells in the absence of any serum?
There is no serum in the L-15 media. We do not continue to culture the cells here as we only check for initial yields and viability with trypan blue. If you intend to culture the cells for a prolonged period serum would be beneficial.
Unfortunately we do not culture the cells past the initial viability and cell countings the choice of media and supplements is a question that would be best directed to media specialists (i.e. JRL or Gibco) or by checking references. Some of the references listed below may be helpful to you in your choice of media and supplements;
http://www.nature.com/cdd/journal/v8/n4/full/4400810a.html
http://www.biomedcentral.com/1471-2121/5/15
http://circ.ahajournals.org/cgi/content/full/100/23/2373#SEC1
Can the Neonatal Cardiomyocyte Isolation System (NCIS Kit) be used on adults?
The NCIS kit is specifically packaged for five separate tissue dissociations, each containing up to twelve neonatal rat hearts. The NCIS protocol is available online here. Adult rat cardiomyocytes usually require perfusion ie. Langendorf Perfusion System and Collagenase Type II.
What are the volumes of Hank's, Earle's and Leibovitz L-15 Media that you sell?
Sterile calcium and magnesium free Hank's Balanced Salt Solution, p 7.4 is supplied in the NCIS (Neonatal Cardiomyocyte Isolation System) kit. A 500ml bottle may be purchased, catalog #LK003210.
The NCIS kit also contains a pouch of Leibovitz L-15 Media Powder. Reconstitute the entire contents of the pouch into a beaker containing 800ml of cell culture grade water. Rinse pouce 2-3 times with additional 100ml. Bring total volume to 1 liter and filter through a 0.22 micron pore size filter. The pouch containing Leibovitz L-15 Media powder for 1 liter is catalog #LK003250.
Sterile Earle's Balanced Salt Solution (EBSS) with calcium, magnesium, bicarbonate and phenol red is supplied with the PDS (Papain Dissociation System) kit. A 100 ml bottle may be purchased, catalog #LK003182.
What is an acceptable enzyme diluent for Neutral Protease/Dispase (code NPRO/NPRO2)?
Neutral protease is usually dissolved in neutral balanced salt solution of choice (ie PBS, Hanks, Earles etc). The composition of several balanced salt solutions are posted in our Tissue Dissociation Guide at http://www.worthington-biochem.com/tissuedissociation/working.html
Usual working solutions for Neutral Protease are 0.5-3.0 u/ml but this varies widely and is dependent on application, diluent, incubation time and temperature. We generally recommend diluting the enzyme as needed as the lyophilized powder is stable at 2-8C. Reconstituted enzymes should not be stored at 2 - 8°C. If necessary they can be aliquoted and frozen at -20°C. Avoid repeated freeze-thaw cycles.
What are acceptable enzyme diluents for Deoxyribonuclease (D/DPFF/DPRF)?
D/DPFF: May be dissolved in 5mM acetate, 1 mM calcium, pH 4.5 and stored in single use aliquots at -20 or -70 for 1 yr.
DPRF: Use water or any buffer pH 4-9 except phosphate, avoid calcium chelators, add 50% glycerol for storage as liquid at -20C. Aliquot avoid repeated freeze/thaw cycles.
Special care must be taken with deoxyribonuclease. Mix gently to avoid shear denaturation. In general, Dnase I is activated by bivalent metals. Maximum activation is attained with Mg+ and Ca+. Dnase I is inhibited by EDTA and SDS. The optimum pH of Deoxyribonuclease I from Bovine Pancreas is 7.8.
What is an acceptable enzyme diluent for Elastase (ES)?
The choice of buffer, concentration and pH are usually dependent on application. Elastase is relatively hydrophobic so solubility at concentrations greater than 0.25% and neutral pH are challenging.
Elastase is stable in the pH range 4-10.4. The optimium pH is near 8.5. For cell culture applications where the material is dissolved in neutral balanced salt solutions and higher concentrations (>2.5%) are desired, increasing the pH to 9-10 aids in solubility and then pH can be lowered to neutral to keep cells happy.
Most labs are dissolving directly in balanced salt solution of choice. It is always best to make a fresh solution as needed but if you must store stocks you can aliquot and freeze at -20C or -80C. You would have to determine frozen stability with your conditions. Elastase should be sterile filtered through a 0.22um pore size membrane before use.
What is an acceptable enzyme diluent for Papain (PAP/PAPL)?
A common working solution of papain is 20 u/ml papain in 1mM L-cysteine with 0.5mM EDTA dissolved in balanced salt solution of choice (ie EBSS etc). EDTA, cysteine and dimercaptoethanol are considered stabilizers. Optimum pH range is 6.0-7.0.
What is an acceptable enzyme diluent for Trypsin (TRL)?
Stock solutions of trypsin should be made initially by reconstituting in 0.001N HCL. This solution can be diluted in the digestion medium or buffer immediately prior to use. The optimum pH of Trypsin is approximately 8.0.
What are conversion factors for units of Collagenase (CLS)?
Unfortunately there is no consistent conversion unit between Wunsch and Mandl units.
Worthington assay is based on modified Mandl unit approx. 0.15 Wunsch units equal 200 Mandl units.
What are conversion factors for units of Deoxyribonuclease (D/DP/DPRFF)?
Note: Kunitz units as reported by other suppliers can be 2-4 times higher than Kunitz units as measured at Worthington. As measured at Worthington one Kunitz unit digests 1 ug of calf thymus DNA in 10 min at 37C in 50mM Tris, 1 mMMg++, 1 mMCa++, pH 7.8. Correlation of digestion units with Kunitz units is different for other DNA and buffer systems. When protease is present, units can not be correlated.
What are conversion factors for units of Trypsin (TRL)?
1 mg Worthington TRL> 180 TAME units >10,350 BAEE units >3,450 USP/NF units
What is the enzyme digestion scale?

The above scale shows the relative digestive power of the enzymes commonly used for tissue dissociation.
What are the basic differences among the four types of collagenase?
| CLS-1 | Contains average amounts of assayed activities (collagenase, caseinase, tryptic and clostripain). Recommended for epithelial, liver (hepatocytes), lung, fat (adipocytes) and adrenal cell isolations. |
| CLS-2 | Contains greater proteolytic activities, especially clostripain activities. It is generally used for heart (cardiomyocytes), bone, muscle, thyroid, cartilage and endothelial cells. |
| CLS-3 | All activities lower than average. Low caseinase and low proteolytic activity. It is usually used for mammary cells and other soft tissues. |
| CLS-4 | Selected because of low tryptic activity. High collagenase required. It is commonly used for pancreatic islets and other applications where receptor integrity is crucial. |
What are the test specifications for each type of collagenase?
| collagenase | caseinase | clostripain | tryptic | A280 | |
|---|---|---|---|---|---|
| CLS 1 | ≥125 u/mg | ≥200 u/mg | ≤4.0 u/mg | ≤0.5 u/mg | report |
| CLS 2 | ≥125 u/mg | ≥200 u/mg | ≥3.5 u/mg | ≥0.1 u/mg | report |
| CLS 3 | ≥100 u/mg | ≥50 u/mg | ≤3.0 u/mg | ≤0.3 u/mg | report |
| CLS 4 | ≥160 u/mg | ≥100 u/mg | ≤3.0 u/mg | ≤0.1 u/mg | report |
| CLSPA | ≥500 u/mg | ≤50 u/mg | ≤2.0 u/mg | ≤0.25 u/mg | report |
Does Collagenase Type I include Brain Heart infusion (BHI)?
Brain Heart Infusion (BHI) fermentation media has not used in the the production of Worthington CLS-1 or any Worthington collagenase product.
What types of media work best with Collagenase I? A colleague of mine uses DMEM but we always use PBS with either 2% FBS or 0.5% BSA. Do you have any suggestions of what might be the best media? We are using it for mouse bone marrow isolation.
Collagenase works well in most balanced salt solutions (ie. Hanks, Earles, PBS, DMEM). The choice is usually more application or tissue specific. We are not working with murine bone marrow here, so I hesitate to choose one over the other. Suggestions are usually based on references. You can search our Tissue Dissociation Guide at www.tissuedissociation.com for references by tissue type.
What is the expiration time for the neonatal cardiomyocyte isolation system?
The NCIS kits are given one year dating from initial release/assay. You can always request information on reassay date prior to purchase.
Previously I've used Dispase II at around 2.5 mg/ml at 37C for 2hr or overnight at 4C to dissociate epidermis from dermis in skin biopsies. What concentration of your neutral protease do you recommend for this procedure?
Dispase II usually refers to a partially purified grade of dispase
similiar to Worthington's NPRO2. Historically most lots of NPRO2
average around 0.4 u/mg so 1u/ml of our neutral protease should be
similar to 2.5 mg Dispase II. Neutral protease working concentrations
are usually in the 0.5-2.0 u/ml range.
Can I use the Neonatal Cardiomyocyte Isolation System with mice?
The Neonatal Cardiomyocyte Isolation System is specifically packaged and use tested on neonatal rats.
A few researchers have used the system on neonatal mice, with some modifications. I have copied some references below for neonatal mice. It seems neonatal mouse hearts may require higher enzyme concentrations and/or longer digestion times than the rats. Increasing the collagenase digestion time to 45 min is a good place to start your optimization. Also one researcher has informed me he increased the trypsin concentration to 100 ug/ml by cutting final volumes in half.
http://ajpcell.physiology.org/cgi/content/full/277/6/C1277#SEC1
http://www.jbc.org/cgi/content/full/277/50/48282
http://www.pnas.org/cgi/content/full/95/16/9669#MaterialsMethods
The protocol for the NCIS kit can viewed by clicking here.
How can I convert units/mg dw of hyaluronidase into U/ml?
Worthington supplies hyaluronidase as a lyophilized powder reporting the u/mg dry weight on the Certificate of Analysis. You can resuspend hyaluronidase to the desired u/ml using this information.
As an example, the current lot of hyaluronidase was released at 525 u/mg dw, if you need a 500 u/ml solution you would dilute 5 mg with 5.25 ml to obtain the 500 u/ml. Calculation 5mg*525u/mg=2625 units/5.25 ml=500 u/ml
How do the different types of collagenase offered by Worthington compare to collagenase from other suppliers?
Worthington Collagenase Cross Reference Chart
| Worthington CLS-1 | Worthington CLS-2 | Worthington CLS-3 | Worthington CLS-4 |
|---|---|---|---|
| Invitrogen/Gibco I | Invitrogen/Gibco II | Invitrogen/Gibco III | Invitrogen/Gibco IV |
| Roche Collagenase A | Roche Collagenase B | Roche Collagenase D | |
| Sigma Type I | |||
| USB type I |
Are your collagenase products tested for endotoxin?
Worthington has instituted endotoxin testing of collagenase. The Limulus amebocyte lysate (LAL) test is used for the quantitation of endotoxin levels in samples. Results are reported on Certificate of Analysis.
I ordered Collagenase IV from your company (100mg). Some of the protocols call for 2mg/ml concentration and others call for 0.2% solution. How does one prepare a 0.2% solution?
A 0.2% solution is equivalent to 2 mg/ml. Just remember a 1% solution refers to 1 gram per 100 ml or 10 mg/ml.
Which type of collagenase is present in your purified collagenase, CLSPA?
CLSPA is bacterial collagenase from clostridium histolyticum that is further purified over a column and pooled based on activity. CLSPA has higher collagenase activity (>500 u/mg DW but usually around 1000 u/mg) and significantly lower caseinase, clostripain and tryptic activity than our cruder CLS types 1-4. We do not characterize the specific composition of each lot but it would be a mixture of several collagenases containing both collagenase A and B (sometimes referred to as I and II). A daily listing of Collagenase activities including CLSPA is posted here.
We ordered hyaluronidase from worthington cat no LS005474. the activity says that it is >/3000 USP/NF units per mg dry weight. Can you please convert it to mg. thanks
Hyaluronidase is sold on a unit basis. The mg supplied may vary slightly between lots as determined by the u/mg activity of each lot. The current lot of HSEP LS005474 is 58E10498 which was released at 7,654 u/mg dry weight. For this lot the 30,000 unit package will contain ~4 mg (30,000 units/7654 u/mg=4mg). If you have a different lot number of HSEP you can check the Certificate of Analysis from the following link- http://www.worthington-biochem.com/index/technical.htmlI have a question relating to your "Neonatal Cardiomyocyte Isolation System" protocol. Is there any serum added to your L-15 media provided in the kit or do I need to add some separately? If the serum is already present, would it be possible to culture the cells in the absence of any serum?
There is no serum in the L-15 media. We do not continue to culture the cells here as we only check for initial yields and viability with trypan blue. If you intend to culture the cells for a prolonged period serum would be beneficial.
Unfortunately we do not culture the cells past the initial viability and cell countings the choice of media and supplements is a question that would be best directed to media specialists (i.e. JRL or Gibco) or by checking references. Some of the references listed below may be helpful to you in your choice of media and supplements;
http://www.nature.com/cdd/journal/v8/n4/full/4400810a.html
http://www.biomedcentral.com/1471-2121/5/15
http://circ.ahajournals.org/cgi/content/full/100/23/2373#SEC1
Protein Sequencing
Answer
What are the recommended conditions for proteolytic digestions using Protease S. aureus V8 (Endoproteinase-Glu-C) ?
Protease S. aureus V8 (Endoproteinase-Glu-C) specifically cleaves peptide bonds on the COOH-terminal side of either aspartic or glutamic acids. In the presence of ammonium, the enzyme specificity is preferential to glutamic sites. It has a molecular weight of 27,000 daltons and optimum pH's of 4.0 and 7.8 with hemoglobin as the substrate. Protease S. aureus V8 is inhibited by diisopropylfluorophosphate and monovalent anions such as F-, Cl-, CH3COO- and NO3.
Typically STAP is used at low enzyme/protein ratio ranges 1:10 to 1:50 and incubated for several hours (2-48hrs) at 37C in ammonium buffers (pH range 4-8).
What are the recommended conditions for proteolytic digestions using Protease S. aureus V8 (Endoproteinase-Glu-C) ?
Protease S. aureus V8 (Endoproteinase-Glu-C) specifically cleaves peptide bonds on the COOH-terminal side of either aspartic or glutamic acids. In the presence of ammonium, the enzyme specificity is preferential to glutamic sites. It has a molecular weight of 27,000 daltons and optimum pH's of 4.0 and 7.8 with hemoglobin as the substrate. Protease S. aureus V8 is inhibited by diisopropylfluorophosphate and monovalent anions such as F-, Cl-, CH3COO- and NO3.
Typically STAP is used at low enzyme/protein ratio ranges 1:10 to 1:50 and incubated for several hours (2-48hrs) at 37C in ammonium buffers (pH range 4-8).
Product Support
Answer
Is your Purified Collagenase, product code CLSPA, available as part of the CLS Sampling Program?
No, the sampling program was designed to eliminate the lot to lot variability the of cruder collagenases. With CLSPA there really is no difference lot to lot because most of the other proteases have been significantly removed or reduced with the column purification. If a researcher wants to try CLSPA, he/she would need to purchase the 4KU vial(approximately 4 mg).
The cruder collagenases are still widely used because the "other" proteases in crude collagenase aid in the digestion. Connective tissue is held together but more things than just collagen, so when the purified collagenase is used it is often used in conjunction with another proteases (enzyme cocktail). See http://www.tissuedissociation.com/theory.html and optimizing strategy under http://www.tissuedissociation.com/techniques.html
Answer
What are some guidelines for clostripain usage?
Clostripain cleaves peptides at the C-terminal side of arginine residues, calcium is required for maximium activity. Reducing agents like DTT, cysteine or mercaptoethanol are considered activators as they protect from inactivation by oxidation. The enzyme is inhibited by oxidizing agents, sulfhydryl reactants, and heavy metal ions. Citrate, borate and Tris anions are less inhibitory.
Optimium conditions for use will have to be determined empirically but I have tried to answer general questions below.
1. what protein concentration should I use?
typical ranges of enzyme to sample protein are 1/20 to 1/100
2. How long will it take?
Incubation time is dependent on nature of the the protein to be digested and temperature- typical range- 1 to several hours at 37°C.
3. What buffer to use?
Buffer of choice containing calcium 1mM and 2.5mM DTT (or mercaptoethanol) pH range 7.4-7.8
Can initially reconstitute clostripain in water at 1 mg/ml aliquot and freeze(avoid repeated freeze thaw cycles)
The following references/link are using clostripain for protein cleavage and may help you develop a protocol:
http://www.jbc.org/cgi/content/full/272/42/26530#SEC1
http://www.jbc.org/cgi/content/full/278/34/31796#SEC1
Answer
What strain of HIV is your recombinant HIV reverse transcriptase (HIVRT) from?
Worthingtons HIVRT was cloned from the subclone of HIV designated pHX2D. This is a subclone of HIV type 1 strain HXB2 -GenBank accession #K03455(M38432). The sequence of HXB2 is described in, Rather et al, 1985, Nature 313:277-284. The source is E. coli plasmid pRC-RT.
Cloning of reverse transcriptase for HIV pHX2D is described in the following papers:
Becerra, S.P, Kumar, A and Wilson, SH, Protein Expression and Purification 4:187-199, 1993.
Becerra et al, Biochemistry 30:11707-11719, 1991.
Our recombinant clone expresses the p66/p51 heterodimer which is representative of the native, wild type protein(accession #V53871). .
Answer
How much endotoxin is contained in your Ovalbumin?
Endotoxin testing is not a release criteria for ovalbumin codes OA (LS003049) or OAC (LS003054) so most lots are not tested. If you are looking for low endotoxin ovalbumin you would need code OAEF (LS003059,3061,3062) where endotoxin values are less than 1 EU/mg
Answer
I purchased phosphodiesterase I as a powder. I need to weight very small amounts of powder in each experiment and I prefer to dissolve it in a buffer and keep it like that. Is it O.K to do so? and if so, in what buffer?
Our QC department reconstitutes the 100 unit vial of phosphodiesterase I (code VPH) with 2ml of 0.11M Tris pH 8.9 containing 0.11M NaCl and 15 mM MgCl, aliquots (to avoid repeated freeze/thaw cycles) and freezes at -20°C. Under these conditions the frozen aliquots are stable for several months
Answer
I bought Deoxyribonuclease I (DCLS) from your company. How can I make the stock solution? Can I disslove the powder in distilled water?
Depending on the application DNase I can be dissolved in water or numerous buffers. Once reconstituted the material should be aliquoted and frozen at -20°C or -80°C. Avoid repeat freeze/ thaw cycles. For long term storage we dissolve Dnase I in 5mM acetate buffer pH 4.5 containing 1mM calcium. It is best to freeze stocks near 1mg/ml as very dilute solutions will not be as stable.
Answer
Is ribonuclease A (RAF, filtered) still stable after being stored at -20°C (product says to store at 2-8)?
Ribonuclease A can be stored frozen at -20°C. We store lyophilized ribonuclease A (code RAF) at 2-8°C and it is stable for at least two years. Storing ribonuclease A at -20°C will not harm the enzyme.
Answer
We ordered hyaluronidase from worthington cat no LS005474. the activity says that it is >/3000 USP/NF units per mg dry weight. Can you please convert it to mg. thanks
Hyaluronidase is sold on a unit basis. The mg supplied may vary slightly between lots as determined by the u/mg activity of each lot. The current lot of HSEP LS005474 is 58E10498 which was released at 7,654 u/mg dry weight. For this lot the 30,000 unit package will contain ~4 mg (30,000 units/7654 u/mg=4mg). If you have a different lot number of HSEP you can check the Certificate of Analysis from the following link- http://www.worthington-biochem.com/index/technical.html
Is your Purified Collagenase, product code CLSPA, available as part of the CLS Sampling Program?
No, the sampling program was designed to eliminate the lot to lot variability the of cruder collagenases. With CLSPA there really is no difference lot to lot because most of the other proteases have been significantly removed or reduced with the column purification. If a researcher wants to try CLSPA, he/she would need to purchase the 4KU vial(approximately 4 mg).
The cruder collagenases are still widely used because the "other" proteases in crude collagenase aid in the digestion. Connective tissue is held together but more things than just collagen, so when the purified collagenase is used it is often used in conjunction with another proteases (enzyme cocktail). See http://www.tissuedissociation.com/theory.html and optimizing strategy under http://www.tissuedissociation.com/techniques.html
What are some guidelines for clostripain usage?
Clostripain cleaves peptides at the C-terminal side of arginine residues, calcium is required for maximium activity. Reducing agents like DTT, cysteine or mercaptoethanol are considered activators as they protect from inactivation by oxidation. The enzyme is inhibited by oxidizing agents, sulfhydryl reactants, and heavy metal ions. Citrate, borate and Tris anions are less inhibitory.
Optimium conditions for use will have to be determined empirically but I have tried to answer general questions below.
1. what protein concentration should I use?
typical ranges of enzyme to sample protein are 1/20 to 1/100
2. How long will it take?
Incubation time is dependent on nature of the the protein to be digested and temperature- typical range- 1 to several hours at 37°C.
3. What buffer to use?
Buffer of choice containing calcium 1mM and 2.5mM DTT (or mercaptoethanol) pH range 7.4-7.8
Can initially reconstitute clostripain in water at 1 mg/ml aliquot and freeze(avoid repeated freeze thaw cycles)
The following references/link are using clostripain for protein cleavage and may help you develop a protocol:
http://www.jbc.org/cgi/content/full/272/42/26530#SEC1
http://www.jbc.org/cgi/content/full/278/34/31796#SEC1
What strain of HIV is your recombinant HIV reverse transcriptase (HIVRT) from?
Worthingtons HIVRT was cloned from the subclone of HIV designated pHX2D. This is a subclone of HIV type 1 strain HXB2 -GenBank accession #K03455(M38432). The sequence of HXB2 is described in, Rather et al, 1985, Nature 313:277-284. The source is E. coli plasmid pRC-RT.
Cloning of reverse transcriptase for HIV pHX2D is described in the following papers:
Becerra, S.P, Kumar, A and Wilson, SH, Protein Expression and Purification 4:187-199, 1993.
Becerra et al, Biochemistry 30:11707-11719, 1991.
Our recombinant clone expresses the p66/p51 heterodimer which is representative of the native, wild type protein(accession #V53871). .
How much endotoxin is contained in your Ovalbumin?
Endotoxin testing is not a release criteria for ovalbumin codes OA (LS003049) or OAC (LS003054) so most lots are not tested. If you are looking for low endotoxin ovalbumin you would need code OAEF (LS003059,3061,3062) where endotoxin values are less than 1 EU/mg
I purchased phosphodiesterase I as a powder. I need to weight very small amounts of powder in each experiment and I prefer to dissolve it in a buffer and keep it like that. Is it O.K to do so? and if so, in what buffer?
Our QC department reconstitutes the 100 unit vial of phosphodiesterase I (code VPH) with 2ml of 0.11M Tris pH 8.9 containing 0.11M NaCl and 15 mM MgCl, aliquots (to avoid repeated freeze/thaw cycles) and freezes at -20°C. Under these conditions the frozen aliquots are stable for several months
I bought Deoxyribonuclease I (DCLS) from your company. How can I make the stock solution? Can I disslove the powder in distilled water?
Depending on the application DNase I can be dissolved in water or numerous buffers. Once reconstituted the material should be aliquoted and frozen at -20°C or -80°C. Avoid repeat freeze/ thaw cycles. For long term storage we dissolve Dnase I in 5mM acetate buffer pH 4.5 containing 1mM calcium. It is best to freeze stocks near 1mg/ml as very dilute solutions will not be as stable.
Is ribonuclease A (RAF, filtered) still stable after being stored at -20°C (product says to store at 2-8)?
Ribonuclease A can be stored frozen at -20°C. We store lyophilized ribonuclease A (code RAF) at 2-8°C and it is stable for at least two years. Storing ribonuclease A at -20°C will not harm the enzyme.
We ordered hyaluronidase from worthington cat no LS005474. the activity says that it is >/3000 USP/NF units per mg dry weight. Can you please convert it to mg. thanks
Hyaluronidase is sold on a unit basis. The mg supplied may vary slightly between lots as determined by the u/mg activity of each lot. The current lot of HSEP LS005474 is 58E10498 which was released at 7,654 u/mg dry weight. For this lot the 30,000 unit package will contain ~4 mg (30,000 units/7654 u/mg=4mg). If you have a different lot number of HSEP you can check the Certificate of Analysis from the following link- http://www.worthington-biochem.com/index/technical.html