Phosphoenolpyruvate Carboxylase - Manual
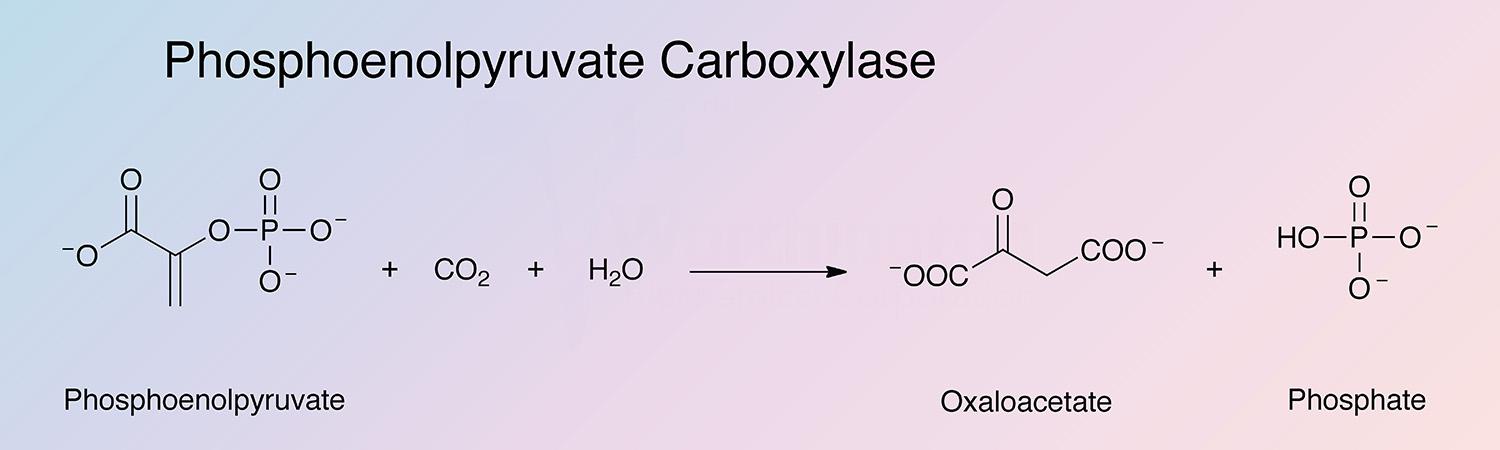
Phosphoenolpyruvate carboxylase (PEPC) catalyzes the fixation of carbon dioxide with phosphoenolpyruvate to produce oxaloacetate and inorganic phosphate.
PEPC is found in most plants and bacteria. It has been reviewed by Utter and Kolenbrander (1972).
Characteristics of PEPC from E. coli:
Apparently only phosphoenolpyruvate and carbon dioxide can participate in the reaction.
The enzyme is a tetramer composed of identical subunits of about 100,000 molecular weight (Wohl and Markus 1972; Yoshinaga et al. 1970). There are thirty-six cysteine residues per mole which exist as free SH groups. See also Izui (1973) for conformation studies and Izui et al. (1970) on active site.
Approximately 400,000 (Smith 1971).
8-9. pH 8.5 is generally used.
Acetyl CoA and fructose-1,6-diphosphate (Utter and Kolenbrander 1972). Acetyl CoA provides maximum activation at about 1 mM concentration, but we were unable to detect significant effects with the fructose diphosphate, guanosine diphosphate, or even the aspartate. Dioxane is very effective, and is used in the assay. However, dioxane generally contains traces of peroxides which must be removed by the dithioerythritol (DTE).
The enzyme is less active in higher ionic strength solutions, showing about 50% activity in 0.15 M ammonium sulfate or potassium chloride. See also Wohl and Markus (1972).
L-aspartate, fumarate and L-malate. (See Izui et al. 1970).
Solutions are stable at least six hours in a Mg2+ buffer. In dilute solution unless Mg2+ or aspartate is present the enzyme dissociates into monomer and dimers and is inactivated Smith and Perry 1973).