For our international customers, please be advised that orders cannot be placed through our website by customers in countries with International Distributor representation.
Introduction to Enzymes
Substrate Concentration
It has been shown experimentally that if the amount of the enzyme is kept constant and the substrate concentration is then gradually increased, the reaction velocity will increase until it reaches a maximum. After this point, increases in substrate concentration will not increase the velocity (delta A/delta T). This is represented graphically in Figure 8.
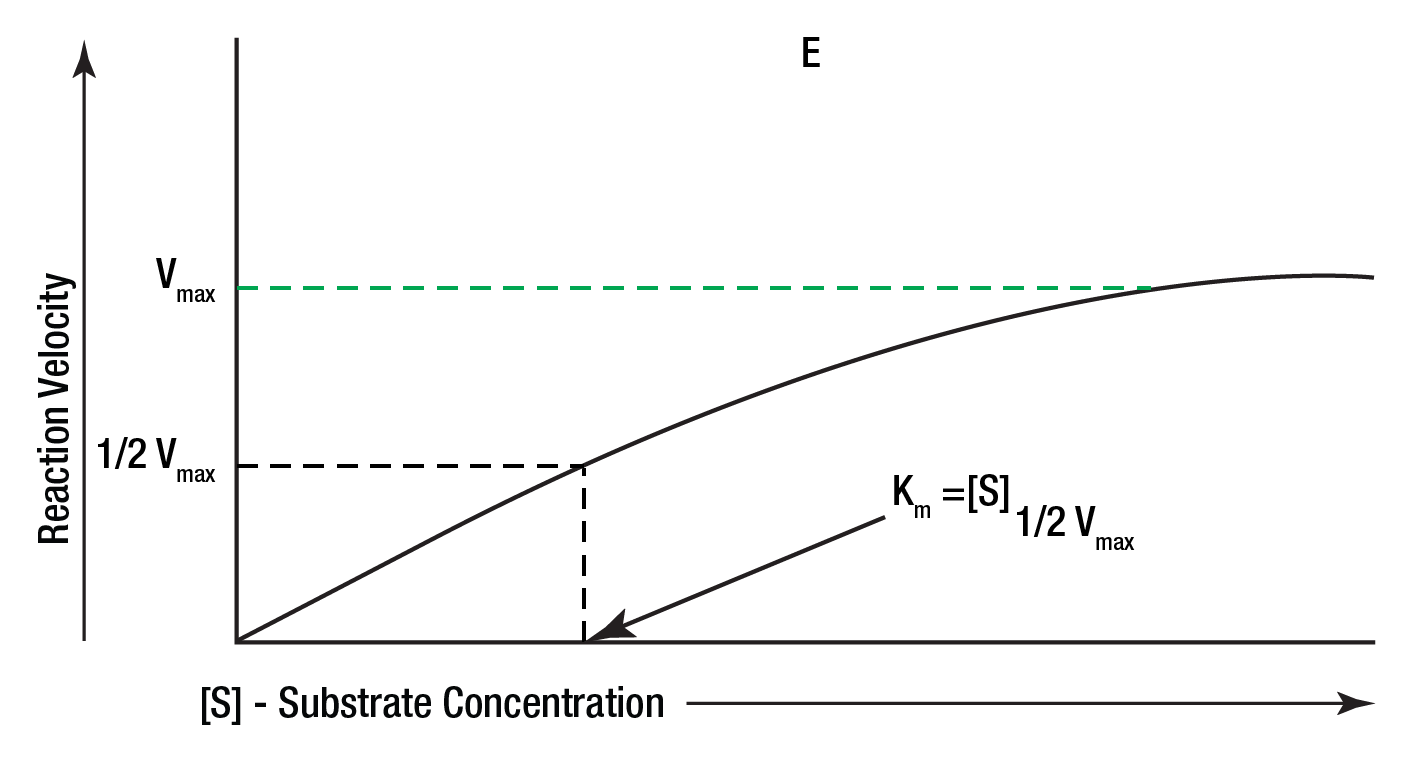
Figure 8: Effect of substrate concetration.
It is theorized that when this maximum velocity had been reached, all of the available enzyme has been converted to ES, the enzyme substrate complex. This point on the graph is designated Vmax. Using this maximum velocity and equation (7), Michaelis developed a set of mathematical expressions to calculate enzyme activity in terms of reaction speed from measurable laboratory data.
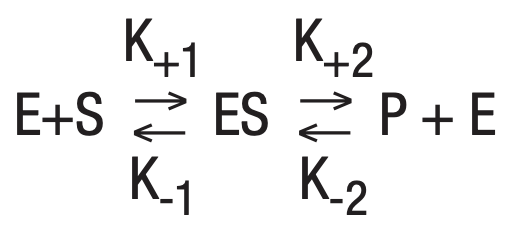 [7]
[7]
The Michaelis constant Km is defined as the substrate concentration at 1/2 the maximum velocity. This is shown in Figure 8. Using this constant and the fact that Km can also be defined as:
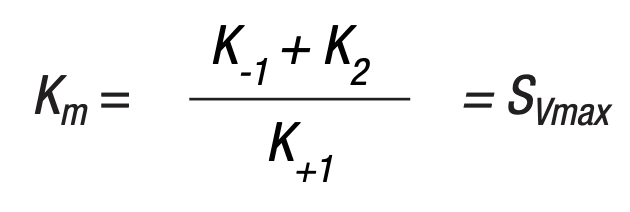
K+1, K-1 and K+2 being the rate constants from equation (7). Michaelis developed the following
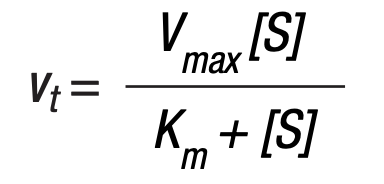
- vt = the velocity at any time
- [S] = the substrate concentration at this time
- Vmax = the highest velocity under this set of experimental conditions (pH, temperature)
- Km = the Michaelis constant for the particular enzyme being investigated
Michaelis constants have been determined for many of the commonly used enzymes. The size of Km tells us several things about a particular enzyme.
- A small Km indicates that the enzyme requires only a small amount of substrate to become saturated. Hence, the maximum velocity is reached at relatively low substrate concentrations.
- A large Km indicates the need for high substrate concentrations to achieve maximum reaction velocity.
- The substrate with the lowest Km upon which the enzyme acts as a catalyst is frequently assumed to be enzyme's natural substrate, though this is not true for all enzymes.