For our international customers, please be advised that orders cannot be placed through our website by customers in countries with International Distributor representation.
Phosphatase, Alkaline - Manual
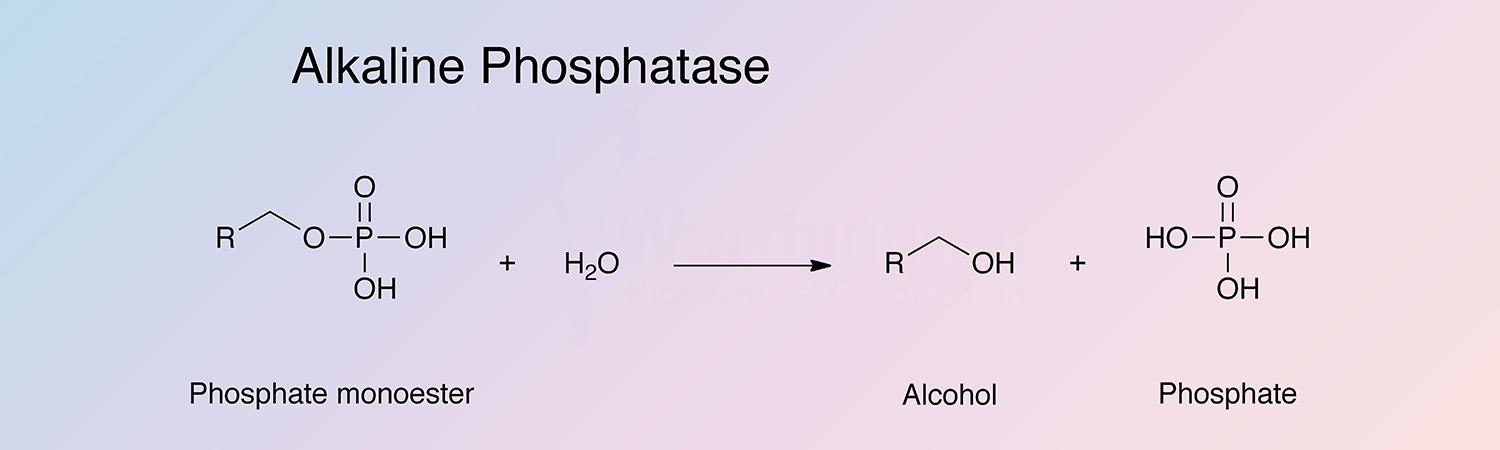
Orthophosphoric-monoester phosphohydrolase (alkaline optimum)
Alkaline phosphatase is a broad term associated with non-specific phosphomonesterases with activity optima at alkaline pH.
The mammalian alkaline phosphatases have been reviewed by Fernley (1971).
E. coli alkaline phosphatase is a valuable reagent for removal of terminal monoesterified phosphate from both ribo- and deoxyribo-oligonucleotide. It has been reviewed by Reid and Wilson (1971).
Characteristics of Alkaline Phosphatase from Chicken Intestine:
This enzyme was partially purified and studied by Kunitz (1960).
Composition: The enzyme is a zinc metallo-enzyme. Schüssler (1968) reports four isozymes. Chang and Moog (1972) found three isozymes in the enzyme from chicken duodenum. (The Worthington preparation is from the whole intestine.)
Optimum pH: 8 - 9.
Activators: Schüssler (1968) indicates activation by Mg2+. See Sivanaesan et al. (1991).
Inhibitors: Acidification to pH 4.5 reversibly inactivates the enzyme.
Stability: The lyophilized preparation is stable for 1-2 years at 2 - 8°C.
Characteristics of Alkaline Phosphatase from E. coli :
Molecular weight: 80,000 Taylor and Coleman (1972). 86,000 Lazdunski and Lazdunski (1969). 89,000 Anderson et al. (1975).
Composition: E. coli alkaline phosphatase is a dimeric, zinc and magnesium containing protein (Anderson et al. 1975; Bosron et al. 1975). Though the subunits are believed to be coded by the same gene (Garen and Garen 1963), they develop molecular heterogeneity after translation (Bosron and Vallee 1975; Bridgen and Secher 1973). See also Schlesinger (1974), Halford et al. (1972). For amino acid composition see Reid and Wilson (1971) and Kelley et al. (1973).
Fernley (1973) and Chappelet-Tordo et al. (1974) indicate there to be two active sites, only one is functional at a time. Two Zn2+ are needed for activity. A stable apoenzyme can be reactivated with Zn2+ , Mn, Co, Ni, Cu, Cd and Hg have been substituted for Zn but only Co restores significant activity (Taylor and Coleman 1972). Magnesium does not activate the apoenzyme but enhances activity of the enzyme containing two gram atoms of zinc (Anderson et al. 1975). See also Anderson and Vallee (1975), Lilja et al. (1975), Brown et al. (1974), Daemen and Riordan (1974), Kaden et al. (1972).
Optimum pH: 8.0 (Garen and Levinthal 1960).
Extinction coefficient: E278 = 7.2 (Anderson et al. 1975).
Isoelectric point: pH 4.5 (Garen and Levinthal 1960).
Activity: Bloch and Schlesinger (1974) report on kinetic studies. Bock and Sheard (1975) indicate that the E. coli enzyme binds phosphate tightly over a wide range of pH forming complexes that may be intermediate in the hydrolytic action. See also Bosron and Vallee (1975), Zukin and Hollis (1975), and Chlebowski and Coleman (1974).
Specificity: See review by Reid and Wilson (1971). Also Mushak and Coleman (1972) and Snyder and Wilson (1972).
Inibibitors: The enzyme is inhibited by chelating agents and inorganic phosphates.
Stability: Stable at 2 - 8°C for at least 6 - 12 months.
Characteristics of Alkaline Phosphatase from Calf Intestine:
Molecular Weight: 140,000.
Optimum pH: 9.8.
Isoelectric Point: 5.7.
Activators: Zn, Mg++, Ca++.